Pharmaceutical Organic Chemistry 1 - Unit 5
Syllabus
Carboxylic acids*
Acidity of carboxylic acids, effect of substituents on acidity, inductive effect and qualitative tests for carboxylic acids ,amide and ester Structure and Uses of Acetic acid, Lactic acid, Tartaric acid, Citric acid, Succinic acid. Oxalic acid, Salicylic acid, Benzoic acid, Benzyl benzoate, Dimethyl phthalate, Methyl salicylate and Acetyl salicylic acid
Aliphatic amines*
Basicity, effect of substituent on Basicity. Qualitative test, Structure and uses of Ethanolamine, Ethylenediamine, Amphetamine
Scroll to Download
POC-I
UNIT-5
SYLLABUS
Carboxylic acids: Acidity of carboxylic acids, effect of substituents on acidity, Inductive effect and qualitative texts for carboxylic acids, amide and ester. Structure and uses of acetic acid, Lactic acid, Tartaric acid, Citric acid, succinic acid, Oxalic acid, salicylic acid, Benzoic acid, Benzyl benzoate, Dimethyl phthalate, Methyl salicylate and Acetyl salicylic acid.
Aliphatic amines: Basicity, effect of substituent on basicity. Qualitative test, structure and uses of ethanolamine, ethylenediamine, Amphetamines.
CARBOXYLIC ACIDS
- These are a class/group of organic compounds that contain a carboxyl group ($-COOH$).
- It can either be aliphatic ($R-COOH$) or aromatic ($Ar-COOH$) based on the alkyl or aryl group, attached to carboxylic Carbon.
- They are commonly found in nature and used in various industries, such as pharmaceuticals, food and cosmetics.
STRUCTURE
- Carboxylic acids have the general formula or where is a hydrocarbon chain.
- The carboxyl group ($-COOH$) consists of a carbonyl group ($C=O$) and a hydroxyl group ($-OH$).
- The carboxyl group is polar and can form hydrogen bond with other molecules.
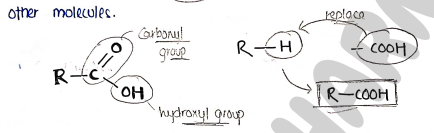
- It lie in one plane and are about 120°.
NOMENCLATURE
It involves two system :-
- Common system Based on the source of acids.
- (Acetic acid) - from vinegar (acetum)
- (formic acid) - from red ant (formica) etc..
- IUPAC System replace suffix with "-oic acid"
- (ethanoic acid) or Methyl Carboxylic acid.
- (methanoic acid).
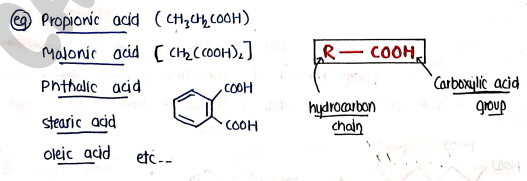
CLASSIFICATION
- Monocarboxylic acids containing one group.
- Dicarboxylic acids containing two groups.
- Polycarboxylic acids which contains more than two groups.
METHOD OF PREPARATION
- By oxidation of Aldehydes
- By oxidation of Alcohols
- By oxidation of Alkenes.
- By hydrolysis of Nitriles
- By hydrolysis of esters
- Carboxylation of Grignard reagents
- Carboxylation of Alkenes (Koch reactions)
1. By oxidation of Aldehydes
- when aldehydes oxidized in the presence of oxidizing agent like potassium permanganate / Nitric acid it produced Carboxylic acid.
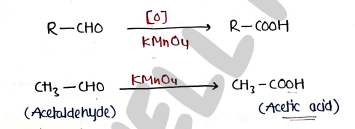
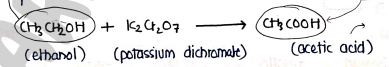
2. By oxidation of primary alcohol ($1^\circ$ alcohol)
- when alcohol oxidized in the presence of oxidizing agent like potassium dichromate ($K_2Cr_2O_7$), it produced carboxylic acid.
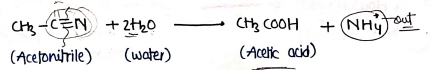
3. By hydrolysis of Nitriles
- when nitriles treated with water/acid catalysis, hydrolysed to produce/form Carboxylic acid.
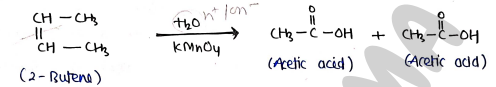
4. By oxidation of Alkenes
- When Alkenes treated with basic potassium permanganate under vigrous conditions, it oxidised to produce carboxylic acids.
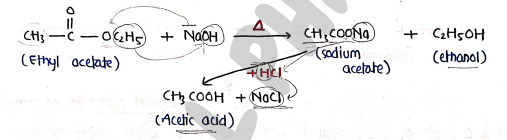
5. By hydrolysis of Esters
- When ester is boiled with concentrated aq. , then treated with acid to produce carboxylic acid.
6. Carboxylation of Grignard reactions
- Grignard reagent ($RMgX$) on treatment with which further on hydrolysis produce carboxylic acids.
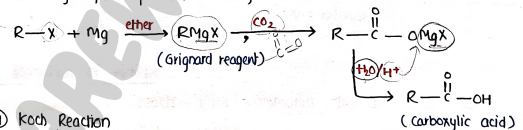
7. Koch Reaction
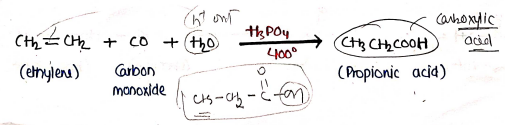
- It is carboxylation of alkenes. The alkene is heated with Carbon monoxide ($CO$) and steam under pressure at 300-400° in the presence of phosphoric acid as the catalyst.
CHEMICAL REACTIONS OF COOH
- Salt formation (Neutralization)
- formation of Acyl halides
- formation of amides
- formation of ester
- formation of anhydrides
1. Salt formation (Neutralization)
- Carboxylic acid reacts with bases to form salts and water.

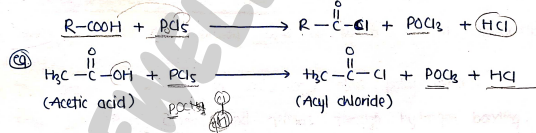
2. formation of Acyl halides
- Carboxylic acid reacts with phosphorus halides ($PCl_5$) to form acyl halides.
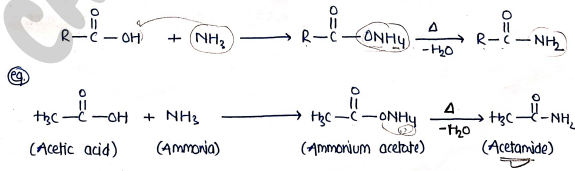
3. formation of Amides
- Carboxylic acid reacts with ammonia to give salt, which on heating produce amides.
4. formation of Ester (Esterification)
- Carboxylic acid react with alcohol in the presence of strong acid to form ester.
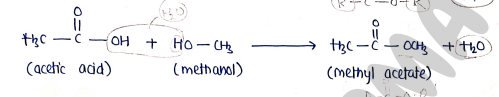
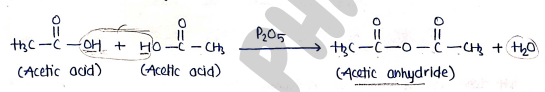
5. formation of Anhydride
- Carboxylic acid undergoes dehydration with phosphorus pentoxide to form acid anhydride.
PHYSICAL PROPERTIES OF COOH
- Carboxylic acids are typically solids or liquids with a characteristics pungent smell & colorless.
- They are soluble in water and other polar solvents.
- They can form dimers and trimers through hydrogen bonding.
- They are weak acids (strong in organic), but can donate a proton ($H^+$ ion) to form conjugate base.
- Boiling & melting point increases with increasing chain length and decreases with increasing branching.
ACIDITY OF CARBOXYLIC ACIDS
- Carboxylic acids are a class of organic compound that contain a carboxyl group (-COOH$) i.e. $R-COOH
- Carboxylic acids are acidic in nature because on dissociation it releases ion/proton.

- Acc. to Arrhenius, those substances who releases ion on dissociation with water are known as Acids.
- So, (Carboxylic acids) are acidic in nature.
- Now, the acidity/stability of substances is depends on that the stability of conjugate base.
- In this, after releasing ion, carboxylic acid form Carboxylate ion.
- "The more stability of carboxylate ion, the more acidity of carboxylic acid"
- Here, Carboxylic acid as well as Carboxylate ion both are Stabilized by Resonance.
- However, carboxylate ion is more stabilized by resonance. so, Carboxylic acids behaves as strong acids.
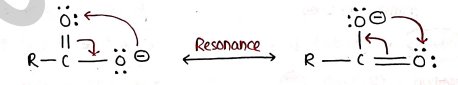
- So, due to resonance they are still stable, after releasing ion even.
OTHER REASONS
- Carboxylic acids are weak acids and the values of Ka for most of aliphatic/aromatic C.A. are lying within range. to (pKa = 4.0-5.0) Acetic acid 4.76
- C.A. are acidic in nature due to that pulls electron towards itself.
Why Carboxylic acids are more acidic than Alcohols
- Lesser the pKa value Acidity
- pKa for Carboxylic acids (4-5) < Alcohols (14-16).
- Greater resonance than alcohols.
EFFECT OF SUBSTITUENTS ON ACIDITY OF C.A.
There are mainly two types of groups which affects the acidity.
1. Electron Withdrawing Groups (-I effect)
- Those who withdraw/receive electron from substance.
- decreases the density.
- increasing stability by withdrawing electron.
- Stability acidity
- Halogens ($F, Cl, Br, I$), , , , Aryl ($Ar$) etc..
2. Electron Releasing/donating Groups (+I effect)
- Those who donate electron to the substance.
- Increases the density.
- decreasing stability by donating electron.
- stability acidity
- Alkyl ($R$), Hydroxyl ($-OH$), Amino ($-NH_2$), Ether ($-OR$), Alkoxy etc..
INDUCTIVE EFFECT
- It is a partial displacement of electrons towards a more electronegative atom of a bond. e.g.
- In this, Cl are more electronegative atom. so,
- It is works/transmitted through several sigma ($\sigma$) bonds.
- It is of two types:-
1. +I effects
- It includes the electron donating/releasing group.
- It release/donate electrons which decreases the stability.
- Basicity, Acidity
2. -I effects
- It includes the electron withdrawing group.
- It withdraw electrons which increases the stability.
- Acidity, Basicity

STABILITY ACIDITY
- EWG acidity of Carboxylic acid
- EDG acidity of Carboxylic acid
QUALITATIVE TESTS
These are those tests which are basically used in the identification of that particular substances.
FOR CARBOXYLIC ACID
Litmus test Carboxylic acids turn blue litmus paper red.
Sodium bicarbonate test When carboxylic acid is added to an aqueous solution of sodium bicarbonate, gas is evolved. $R-COOH + NaHCO_3 \rightarrow R-COONa + CO_2\uparrow + H_2O$
Ester formation test When carboxylic acid on warming with alcohol (ethanol) in the presence of small amount of sulphuric acid, fruity/sweet smell of ester is obtained. $R-COOH + C_2H_5OH \xrightarrow{H^+} R-COOC_2H_5 + H_2O$
FOR ESTERS ($R-C(=O)-OR'$)
Hydroxamic acid test Esters give a purple coloration with hydroxamic acid solution.
KOH test Esters give a yellow coloration with KOH solution.
Bromine water test Esters decolorise bromine water.
FOR AMIDES ($R-C(=O)-NH_2$)
Nitrous acid test Amides gives a red coloration with nitrous acid sol^n.
HCl Solution test Amides gives a white precipitate of ammonium chloride with HCl solution.
KOH solution test Amides do not affect KOH solution.
Bromine water test Amides decolorize bromine water.
STRUCTURE AND USES OF CARBOXYLIC ACIDS
1. ACETIC ACID
- It is a colorless organic liquid compound with a pungent smell and sour taste, commonly found in Vinegar.

- Structure:
- IUPAC: Ethanoic acid
Uses:
- It is used as a food additive, preservative and flavoring agents in foods like pickles, sauces and beverages.
- used in the production of plastic, fibers and dyes.
- also used as cleaning agent and disinfectants in various industries.
2. CITRIC ACID
- It is naturally occuring, weak organic acid found in citrus fruits, with a characteristic sour taste and crystalline appearance.
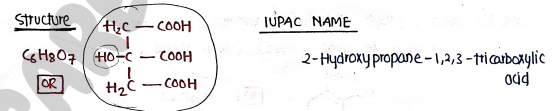
Uses:
- It is widely used as flavoring agent, preservative and acidulant in foods, bevarages and confectionary.
- used as an excipients, buffering agent, intermediate in pharmaceutical applications.
- also used in cosmetics as antioxidants and astringent.
- also used as cleaning agents and disinfectants in various industries.
3. Lactic Acid
- or
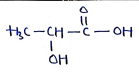
- IUPAC (2-Hydroxypropanoic acid)
Uses: Used as flavoring agent, preservatives, acidulant in foods, beverages and dairy products. Also used in cosmetic products, pharmaceuticals, solvent, cleaning agents etc.
4. Tartaric Acid
- or
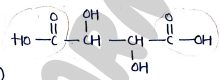
- IUPAC (2,3-dihydroxy butanedioic acid)
Uses: Used as flavoring agent, preservatives, acidulant in foods, bevarages and wine production. also used as excipients, buffering agent, in cosmetics, metal, dyes.
5. Succinic Acid
- or
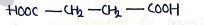
- IUPAC (Butanedioic acid)
Uses: Manufacturing of dyes, volumetric analysis for acid-base titration. also used as flavoring agent, preservatives, buffering, plastic, resins etc.
6. Oxalic Acid
- or
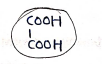
- IUPAC (Ethanedioic acid)
Uses: Redox titration, stain remover, mordant in dyeing, metal polish, allyl alcohol, formic acid (approx. same as above).
7. Salicylic Acid
- (Benzene ring with and at ortho positions)
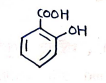
- IUPAC (2-Hydroxybenzoic acid)
Uses: It is used as an analgesic, antipyretic and anti-inflammatory agent for pain relief, fever reduction and skin condition like acne.
- Used as cosmetic, antioxidants, antifungal etc.
- Peeling agent, antidandruff.
- Production of plastic, dyes & other industrial chemicals.
8. Benzoic Acid**
- (Benzene ring with $-COOH$)
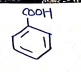
- IUPAC (Benzene carboxylic acid)
Uses: Work as antifungal as well as antimicrobial agent.
- Used in wound cleaning & disinfecting.
- Others. (flavoring, preservatives, cosmetic, Industrial etc.)
9. Acetylsalicylic acid (Aspirin)
- (Benzene ring with and at ortho positions)
Uses:
- Used as Pain reliefer, anti-inflammatory agents to treat conditions like headaches, fever, arthritis.
- Used as an anticoagulant to prevent blood clotting & stroke.
- Also help to prevent cardiovascular events, such as heart attacks or stroke.
10. Dimethyl phthalate
- (Benzene ring with two groups at ortho positions)
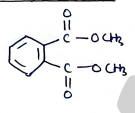
Uses:
- It is used as insect repellent for mosquitos.
- Used as plasticizer in the production of PVC plastic.
- Used as solvent for insecticides, pesticides etc.
11. Benzyl benzoate
- ($C_6H_5-CH_2-O-CO-C_6H_5$)
Uses:
- Used as topical treatment for scabies and lice infections.
- Treatment of acne, eczema dermatitis.
- As antimicrobial and antifungal applications, wound cleaning/disinfecting.
12. Methyl Salicylate
- (Benzene ring with and at ortho positions)
Uses:
- Used as muscle relaxant for pain, arthritis, brushing backaches.
- Relief of muscle & Joint pain, anti-inflammatory.
ALIPHATIC AMINES
SYLLABUS
Basicity, effect of substituent on basicity. Qualitative test, structure and uses of ethanolamine, ethylenediamine, Amphetamines.
INTRODUCTION
Amines: These are derivatives of ammonia ($NH_3$) in which one or more hydrogen atoms have been replaced by alkyl/aryl groups.
Aliphatic Amines: These are a type of organic compound that are derived from Ammonia ($NH_3$) in which one or more hydrogen atoms replaced by alkyl groups.
Replaced by:
- Alkyl group such as etc... (Aliphatic amines)
- Aryl group such as Benzene etc.. (Aromatic amines).
The general formula of aliphatic amines: where is an alkyl group.
examples
- (Methyl amine)
- (Dimethyl amine)
- (Trimethyl amines)
PROPERTIES
- Aliphatic amines are typically colorless liquids or solids with a characteristics odour.
- Aliphatic amines are generally basic in nature. mostly amines.
- They are soluble in water and organic solvents.
- They form salts when reacts with acids. they also reacts with other compounds to form amides and other derivatives.
- IR stretch for N-H is .
CLASSIFICATION
These are classified into three categories :-
Primary amines (1^\circ$) $\rightarrow These are those amines in which one hydrogen atom is replaced by alkyl group in ammonia. e.g. Methyl amines ($CH_3-NH_2$)
Secondary amines (2^\circ$) $\rightarrow These are those amines in which two hydrogen atoms is replaced by alkyl group in ammonia. e.g. Dimethyl amines ($CH_3-NH-CH_3$)
Tertiary amines ($3^\circ$) These are those amines in which three hydrogen atoms are replaced by alkyl group in ammonia. e.g. Trimethyl amines ($(CH_3)_3N$)
NOMENCLATURE
- they are named as Alkylamines or replacing 'e' with amine in IUPAC Alkan-amine.
- Ethyl amine / Ethanamine.
- Ethyl methyl amine / N-Methylethanamine.
BASICITY OF AMINES
Aliphatic amines are the derivatives of ammonia ($NH_3$).
Ammonia are basic in nature which donate lone pair, because it contain lone pair of electrons on Nitrogen.
Aliphatic amines are basic in nature because it contain lone pair so it can donate lone pair of electron.
Acc to Lewis concept, Those substance/elements which accept lone pair are called Acid. Those substances/elements which donate lone pair are called Base.
A substance, which contain lone pair can donate it.
So, Amines are basic in nature and its basicity is depends on those substituents who alter electron density and stability.
The basicity of amines is also depends on electron density (capacity of electrons). electron density Basicity
It is also depends on the stability. stability Basicity
EFFECT OF SUBSTITUENTS ON BASICITY
Basicity of aliphatic amines depend on the nature of substituent.
1. Electron withdrawing Group (-I effect)
- Those groups which withdraw electrons and decrease e- density. e- density = Basicity
- These groups withdraw electrons which increases its stability. stability = Basicity
2. Electron donating Group (+I effect)
- Those groups which donates electron and increases e- density. e- density = Basicity
- These groups donates electrons & decreases its stability. Stability = Basicity
- Alkyl groups ($CH_3, C_2H_5$) etc.
Acc. to this, (Basicity). But this is right for gaseous phase. But due to stearic hindrance in amines (liquid amines), the order becomes .
In case of Aromatic amines:
- Aliphatic amines > Aromatic amines
- Because Aromatic amines have resonance which increases its stability. stability = Basicity.
- Final Basicity Order: 2° Amines > 3° Amines > 1° Amines > Aromatic Amines
QUALITATIVE TESTS FOR AMINES
Solubility test Amines are basic, which react with acid and form salt. (Aniline Anilinium chloride)
Carbylamine test Sample amines + Alc. KOH + Chloroform (2 drops) isocyanide with unpleasant/foul smell.
Diazotisation test of sample + 10 drops of conc HCl. Boil & cool in ice water + 4-5 drops of + \beta$-naphthol in NaOH $\rightarrow observe:
- Yellow solid 2° amines
- Red Color 3° amines
- Orange ppt 1° amines
STRUCTURE AND USES OF AMINES
1. Ethylene diamine ($C_2H_8N_2$)
- It is used in manufacturing of fuel additives, bleach activators, chelating agents, corrosion inhibitors.
2. Amphetamine ($C_9H_{13}N$)
- (Benzene ring)-$CH_2-CH(CH_3)-NH_2$
- It is used as nerve stimulants, it treats attention deficit hyperactivity disorder.
- Act as CNS stimulants.
3. Ethanolamine ($C_2H_7NO$)
- It is used as feedstock in the production of detergents, emulsifier, polishes, corrosion inhibitors.
- used as surfactants, used for removing from natural gas.
METHOD OF PREPARATION
1. Gabriel phthalimide method
- treatment of phthalimide with potassium hydroxide to form the potassium phthalate. Phthalimide + Potassium phthalate ($N-K) + $CH_3CH_2Cl \rightarrow N-ethyl phthalimide + Phthalic acid salt + Ethyl amine ($C_2H_5NH_2$).
2. Reduction of nitroalkane with Ni/Pt
- (Nitroethane ethyl amine)
3. Reaction of alkyl halides with ammonia to form amines.
- Alkyl halides react with alcoholic solution of ammonia to form amines. (Ethyl bromide ethyl amine)
4. Curtis rearrangement
- This reaction converts acyl azide to primary amines.
5. Reduction of oximes
- Primary amines can be obtained by reduction with Ni or Pt. (propionaldehyde oxime n-propyl amine)
REACTIONS
1. Salt formation
- Amines are bases and react with acid to form salt. (ethyl amine ethyl ammonium chloride)
2. Reaction with chloroform and KOH
- Primary amines reacts with chloroform and solution of KOH to form isocyanides. (methyl amine methyl isocyanide)
