Pharmaceutical Organic Chemistry 1 - Unit 4
Syllabus
Carbonyl compounds* (Aldehydes and ketones)
Nucleophilic addition, Electromeric effect, aldol condensation, Crossed Aldol condensation, Cannizzaro reaction, Crossed Cannizzaro reaction, Benzoin condensation, Perkin condensation, qualitative tests, Structure and uses of Formaldehyde, Paraldehyde, Acetone, Chloral hydrate, Hexamine, Benzaldehyde, Vanilin, Cinnamaldehyde.
Scroll to Download
POC-1ST
UNIT-4
CARBONYL COMPOUNDS
ALDEHYDES AND KETONES
SYLLABUS + IMPORTANT QUESTIONS
- Nucleophilic Addition, Electromeric effects, Aldol Condensation, Crossed Aldol Condensation, Cannizaro reactions, Crossed cannizaro reactions, Benzoin condensation, Perkin condensation, Qualitative tests.
- Structure and uses of formaldehyde, Paraldehyde, Acetone, chloral hydrate, Hexamine, Benzaldehyde, Vanillin, Cinnamaldehyde.
INTRODUCTION
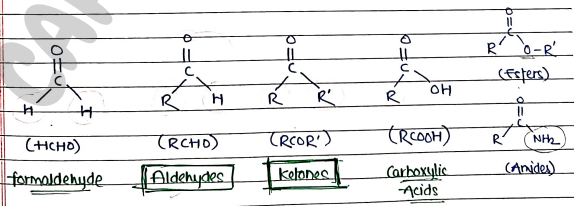
- Carbonyl compound are those organic compounds which contain carbonyl group ($C=O$), which is carbon-oxygen double bond.
- Several compounds are formed from carbonyl group.
Properties
- These are highly reactive compounds.
- They can perform Nucleophilic addition, Electrophilic substitution etc.
- Most carbonyl compounds are polar and soluble in water & organic solvents.
- The carbonyl group has a hybridised carbon and oxygen.
ALDEHYDES
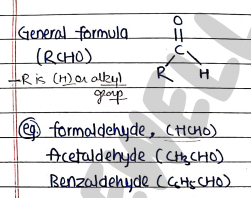
- These are those carbonyl compounds which contain carbonyl group ($C=O$) at the end of carbon chain.
KETONES
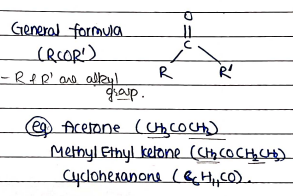
- These are those carbonyl compounds which contain carbonyl group ($C=O$) within the carbon chain.
CHEMICAL REACTIONS OF CARBONYL COMPOUNDS (AFK)
- Nucleophilic Addition Reactions
- Electromeric effect
- Aldol Condensation
- Cross Aldol Condensation
- Cannizaro reactions
- Cross-cannizaro reactions
- Benzoin Condensation
- Perkin Condensation
NUCLEOPHILIC ADDITION REACTIONS
- These are those reactions, in which a nucleophile (a negatively charged or electron rich species) attacks the electrophilic carbonyl carbon of an aldehyde or ketone, resulting in the addition of the nucleophile to the carbonyl group.
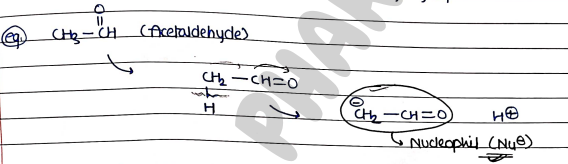
- Both Aldehyde and ketone give nucleophilic addition reactions.
- In normal, one substance act as reagent and other reactant.
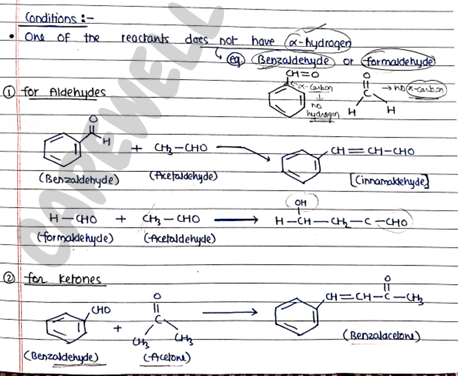
For Aldehydes:
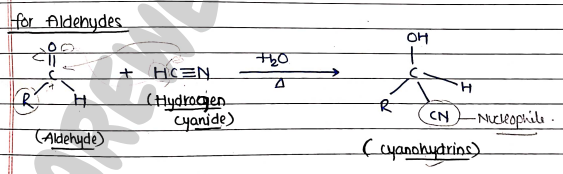
- In this, Aldehyde act as reactant and HCN act as reagent.
- Now, HCN form nucleophile ($CN^-$) and release ion.
- Then this nucleophile ($Nu^-$) attacks on the electrophile of the aldehyde.
- finally, Nu attacked on Carbon (electrophile) atom and ion attached with O and form cyanohydrins.
Mechanism:
- Step 1: protonation of reagent / formation of nucleophile ($Nu^-$)

- Step 2: Attack of nucleophile on reactant.
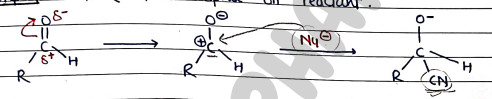
- Step 3: Addition of electrophile ($H^+$)
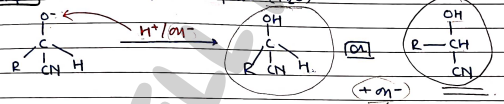
For Ketones:
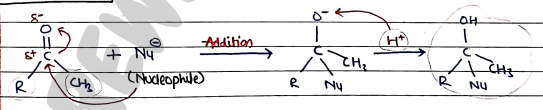
In this, positively polarised carbon atom in act as electrophile and reagent act as nucleophile ($Nu^-$).
firstly, Nu attacks on electrophile and form intermediate which then react with ion and form final product.
Eg: Addition of HCN mechanism is the same as aldehyde.

ELECTROMERIC EFFECTS
- It is a temporary and reversible shift of electron in a multiple bond ($\pi$ bond) toward one of the bonded atom, formed polarised bond.

- Electromeric addition reaction: Shifting of electron and attachment of electrophile ($E^+$) at same side.
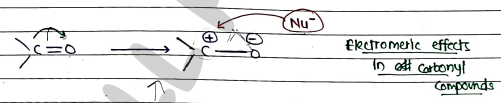
Nucleophilic addition reaction: Sifting of electron and attachment of nucleophile ($Nu^-$) are at different side.
It is of two types:
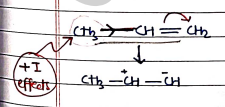
- +E Effects (Positive Electromeric Effects): Electron-withdrawing group (EWG) pull electrons away from the multiple bond, increasing the positivity of adjacent atom.
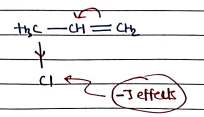
- -E Effects (Negative Electromeric Effects): Electron-donating groups (EDG) push electrons towards the multiple bond, increasing the negativity to adjacent atom.
ALDOL CONDENSATION
- It is a chemical reactions that combines two carbonyl compounds (aldehydes or ketones) to form a new carbon-carbon bond.
Conditions for Aldol Condensation:
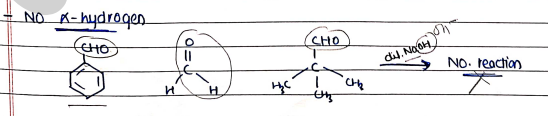
- All aldehydes and ketones, if they have $\alpha$-hydrogen.
- dil. NaOH (for aldehydes), for ketones.
For Aldehydes:

- In this reaction two molecules of aldehyde are condensed together to form Aldol, in the presence of dil. NaOH.
Mechanism:
- Step 1: formation of nucleophile (from reagent).

- Step 2: Attack of Nucleophile on reactant
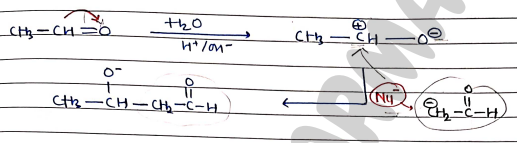
Note : Presence of $\alpha$-hydrogen of aldehyde or ketone is necessary for aldol condensation.
- $\alpha$-hydrogen is necessary to form Nucleophile ($Nu^-$).

- $\alpha$-hydrogen
For Ketones:
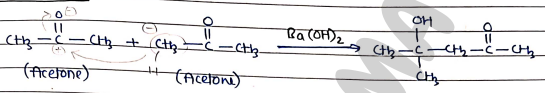
- Some mechanism as aldehyde.
CROSSED ALDOL CONDENSATION
- It involves reaction between two different carbonyl compound (aldehydes or ketones) react to form a new carbon-carbon bond.
Conditions:
- One of the reactants does not have $\alpha$-hydrogen.
Mechanism:
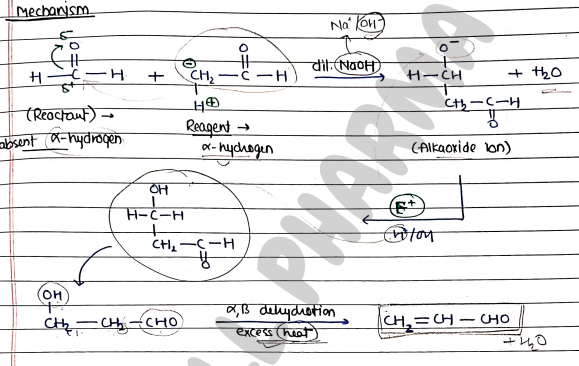
- Step 1: formation of Nucleophile (from reagent).
- Step 2: Attack of nucleophile on reactant.
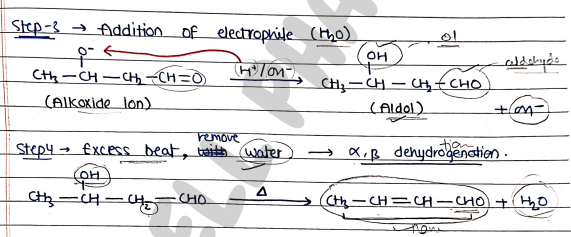
- Step 3: Addition of electrophile ($H_2O$).
- Step 4: removal of through dehydration.
CANNIZZARO REACTIONS
- It is a reaction which involves reaction between two non $\alpha$-hydrogen containing aldehydes.
- They form an alcohol and a carboxylic acid salt in the presence of conc. NaOH (strong base).
Conditions:
- only for aldehydes ($\alpha$-hydrogen absent eg. formaldehyde, Benzaldehyde).
- Involve redox mechanism.
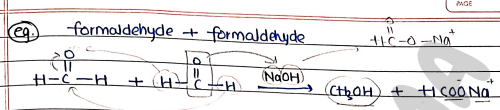
Example: formaldehyde + formaldehyde Methanol (Alcohol) + Sodium formate (Carboxylic acid salt)
Mechanism:
- Step 1: Nucleophilic addition of ion to the 1st aldehyde.
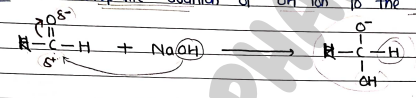
- Step 2: Shifting of hydride ion ($H^-$) from one molecules to other.

- Step 3: formation of alcohol and carboxylic acid salt.
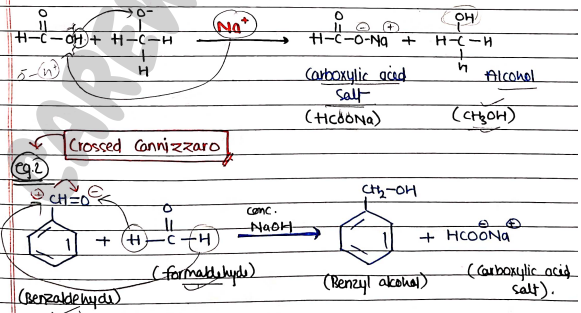
Crossed-Cannizzaro Reactions
- Crossed cannizzaro reactions is a variation of cannizzaro reaction in which two different aldehydes react to form an alcohol and a carboxylic acid salt.
Mechanism:
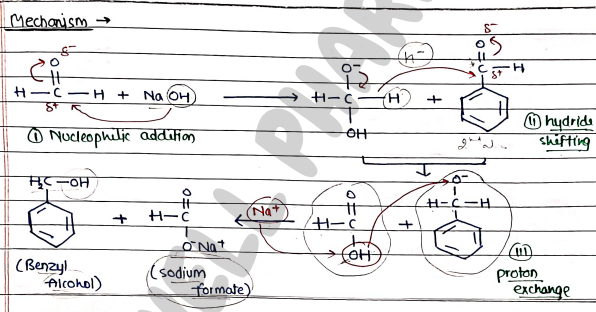
- firstly, ($OH^-$) hydroxide ion attached on the carbon atom and form alkoxide ion.
- shifting of hydride ion from one molecules of aldehyde to another one.
- then transfer of proton from one molecules to another.
- After addition of ion, it form Sodium formate (carboxylic acid salt) and Alcohol (Benzyl alcohol).
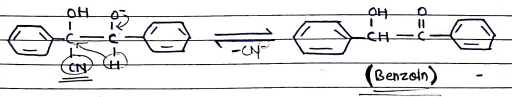
BENZOIN CONDENSATION
- It is a chemical reaction between two molecules of an aromatic aldehyde (typically Benzaldehyde) to form a Benzoin compound.
- It occurs in lack of $\alpha$-hydrogen compounds as aromatic condensation occurs in the presence of potassium cyanide (KCN).
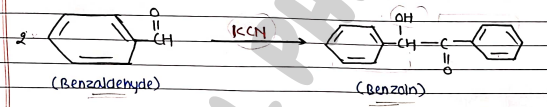
Mechanism:
- Step 1: Cyanide ion react with Benzaldehyde to form Cyanohydrine.
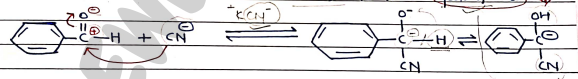
- Step 2: Condensation b/w Cyanohydrin and 2nd molecules of Benzaldehyde.

- Step 3: Rearrangement and Removal of Cyanide ion (forms Benzoin).
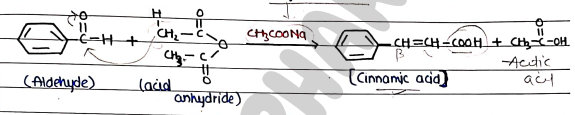
PERKIN CONDENSATION
- It is a reaction which occurs between an aldehyde and acid anhydride to form an $\alpha, \beta$-unsaturated carboxylic acid, in the presence of carboxylic acid salt.
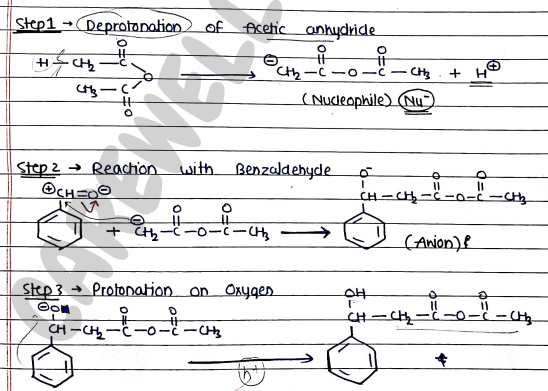
Mechanism:
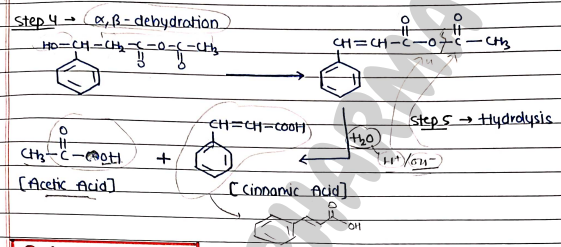
QUALITATIVE TESTS
- Sodium Bisulphite test: when Aldehyde and ketones are react with sodium bisulphite it forms a white precipitate (crystalline compounds).
- 2,4-Dinitrophenylhydrazine (DNPH) test: forms a yellow to orange precipitate with aldehydes and ketones.
- Schiff's test: forms a pink to red color with aldehydes.
- Fehling's test: forms a brick-red precipitate with aldehydes.
- ferric hydroxide test: forms a brown color with ketones.
- Tollen's test: forms a silver mirror with aldehydes.
STRUCTURE AND USES
I. formaldehyde
- It is colorless, strong-smelling gas.
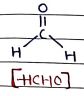
- Uses:
- It is used to produce adhesives, plastic & resins.
- It is used as a disinfectant and preservatives in pharmaceuticals, cosmetics and food industries.
- Also used in cosmetics, soaps, shampoos, lotions etc.
II. Vanillin
- It is a white or very slightly yellow colored needle-like compound.
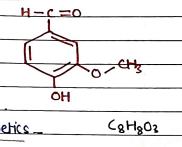
- Uses:
- It is used as a flavouring agent in foods, beverage and pharmaceuticals.
- It is also used in perfumes, fragrances.
- Also used in food additives and cosmetics.
III. Benzaldehyde
- It is a colorless to yellow liquid having a bitter almond odour. It is slightly water soluble but completely insoluble in ethanol and diethyl ether.
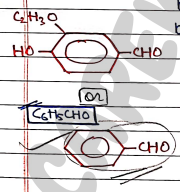
- Uses:
- It is used in perfumery and dye.
- It is used as a solvent for oils, resins, etc.
- Also used as a flavoring agent in cinnamic acid, benzoic acid, and some pharmaceuticals.
- also used as photographic chemicals, baking chemicals, soap.
IV. Acetone
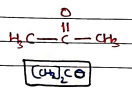
- Uses:
- It is widely used as a solvent in various industries, such as cosmetic, pharmaceutical.
- used for manufacturing plastics, artificial fibers.
- making paper coating, adhesives & heat-seal coating.
V. Paraldehyde
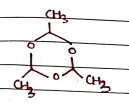
- Uses:
- In ancient time, it was used as sedative and hypnotics medication, but it discontinued due to its toxicity & flammability.
- It has also been used as an anticonvulsant for treating seizures.
VI. Chloral Hydrate ($C_2H_3Cl_3O_2$)
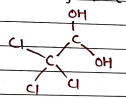
- Uses:
- It is a sedative used in the short-term treatment of insomnia.
- It relieves anxiety and induce sleep before surgery.
- also used in post-surgery as pain killer.
VII. Hexamine ($C_6H_{12}N_4$)
- Methenamine
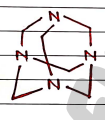
- Uses:
- It is an anti-infective agent used for treating urinary tract infections.
- It also has antibiotic activity.
- also used as a fuel source in camping stoves, emergency rations and military applications.
VIII. Cinnamaldehyde ($C_9H_8O$)
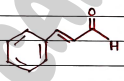
- Uses:
- It have antifungal and antibacterial property.
- It enhance oral health as it fight against tooth decay and bad breath.
- Also used as flavouring agent in liquid refreshment, ice-creams, chewing etc.
- also used in perfumes.
