Pharmaceutical Organic Chemistry 1 - Unit 3
Syllabus
Alkyl halides*
SN1 and SN2 reactions - kinetics, order of reactivity of alkyl halides, stereochemistry and rearrangement of carbocations. SN1 versus SN2 reactions, Factors affecting SN1 and SN2 reactions Structure and uses of ethylchloride, Chloroform, trichloroethylene, tetrachloroethylene, dichloromethane, tetrachloromethane and iodoform.
Alcohols*
Qualitative tests, Structure and uses of Ethyl alcohol, Methyl alcohol, chlorobutanol, Cetosteryl alcohol, Benzyl alcohol, Glycerol, Propylene glycol
Scroll to Download
POC-1ST
UNIT-3
SYLLABUS + IMPORTANT QUESTIONS
Alkyl halides
- and Reactions
- kinetics, order of reactivity, stereochemistry and rearrangement of carbocations.
- VS reactions, factor affecting and reactions.
- Structure and uses of ethyl chloride, chloroform, trichloroethylene, tetrachloroethylene, dichloromethane, tetrachloromethane and iodoform.
Alcohols
- Qualitative tests, structure and uses of ethyl alcohol, Methyl alcohol, chlorobutanol, cetostearyl alcohol, Benzyl alcohol, Glycerol, Propylene glycol.
ALKYL HALIDES
These are those organic compound that contain a halogen atom ($F, Cl, Br, I$) bonded to an alkyl group (a hydrocarbon chain).
General structure:
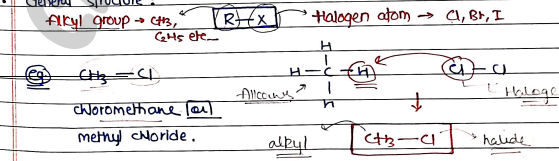
Also known as Haloalkanes or Halogen alkanes.
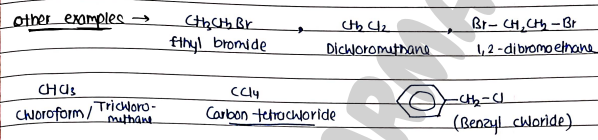
Other examples
Types of Alkyl halides
It is of mainly three types:
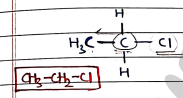
- Primary halides :
- They contains a primary alkyl group bonded to a halogen.
- In this, carbon attached with only one alkyl group/carbon chain, so it is primary halides.
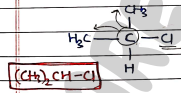
- Secondary halides :
- They contains a secondary alkyl group bonded to a halogen.
- Carbon attached with two other carbon chain/alkyl group, so it is secondary.
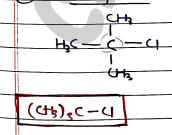
- Tertiary halides :
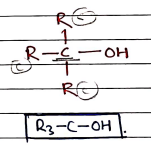
- They contains a tertiary alkyl group ($R_3C-$) bonded to a halogen.
- Carbon attached with three other carbon chain/alkyl group.
Method of Preparations
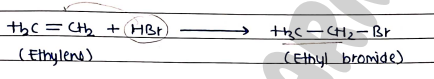
- from Alkenes They can be prepared by the addition of hydrogen halides / halogens to the carbon-carbon double bond.
- Hunsdiecker reactions When silver salt of carboxylic acid react with bromine in the presence of produce Alkyl bromides.

- From alkanes

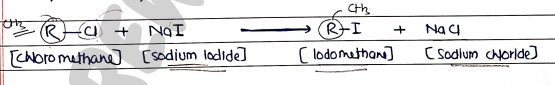
- Halogen exchange (Finkelstein Reactions)
Properties
- Polar covalent bond b/w the alkyl group and halogen due to electrophile nature of halogen.
- Highly reactive towards nucleophilic substitution Reactions ($SN_1$ & $SN_2$).
- Boiling point increases with mol. weight & size.
- Soluble in organic solvents.
AND
SN Nucleophilic Substitution Reactions.
- These are those in which a nucleophile (negatively charged) replaces a leaving group (functional group) in a molecules.
- It form a new covalent bond b/w nucleophile and molecules.
Types
There are two main types of nucleophilic substitution reactions
- (Substitution Nucleophilic Unimolecular)
- (Substitution Nucleophilic Bimolecular)
REACTIONS
- represents Unimolecular Substitution Nucleophilic reactions.
- It is unimolecular so it follow first-order kinetics i.e. it is depends on the concentration of substrate.
- It is a type of reaction, which include two-step mechanism.
- tertiary alkyl halides are more reactive towards this reactions.
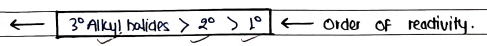
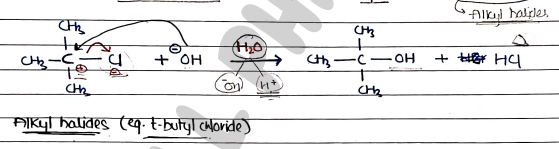
Mechanism
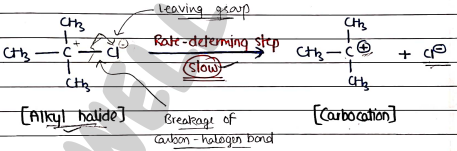
- It is two step mechanism in which first step is rate-determining step.
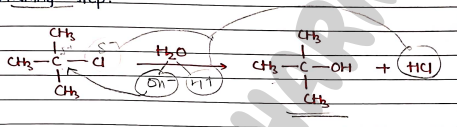
Step 1: It involves the formation of carbocation intermediate. It is slow and rate-determining step.
Step 2: It involves the formation of product in which Nucleophile ($Nu^-$) attacks the carbocation. It is fast.
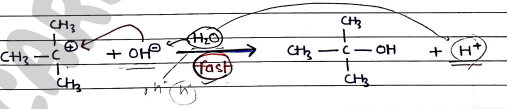
Note:- Since, the first-step is slow and rate-determing, the overall reaction/rate of reaction depends only on the substrate.
Kinetics reaction follow the first-order kinetics, because in reactions the rate of reactions depends only on the concentration of the substrate. Rate (R) = Where:
- R = Rate of reaction
- K = Rate constant
- [RX] = concentration of substrate (Alkyl halides) e.g. If we double the substrate concn, it double the (R).
Order of Reactivity reactions follow
- In reaction, order of reactivity depends on the stability of carbocation.
- Nucleophile attacks on the more stable carbocations.
Factor affecting reactions
- Substrate Tertiary > Secondary > Primary (due to carbocation stability).
- Leaving Group Good leaving group facilitate reactions. .
- Nucleophile weak nucleophile (H_2O, R-OH, NH_3$) favor $SN_1.
- Solvent Polar, protic solvent ($H_2O, ROH$) facilitate reactions.
REACTIONS
- represents Bimolecular Nucleophilic substitution reaction.
- It is Bimolecular, so it follow second order kinetics i.e. it depends on the concentration of both (substrate & Nucleophile).
- It involve one step mechanism.
- Primary alkyl halides are more reactive towards reactions. Order of reactivity:
Mechanism
- It is the single step mechanism, in which nucleophilic attack on the substrate are the rate-determining step. (slow).
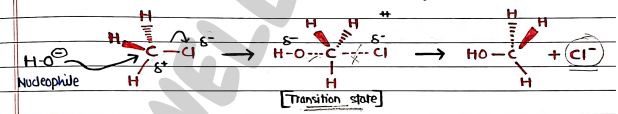
- Firstly Nucleophile ($OH^-$) approaches the substrate from the backside.
- Simultaneous, formation of new bond between the nucleophile and the substrate (partially bonded) and breaking the bond between the substrate and leaving group (partially broken).
- Due to this, transition state formed. It is pentacoordinate, trigonal bipyramidal intermediate.
- At last, chlorine departed/removed and product formed.
Kinetics Second-order kinetics: Rate depends on substrate [S] and Nucleophilic concentration [Nu]. R = where:
- R = rate of reactions.
- K = rate constant
- [S] = concentration of substrate
- [Nu] = concentration of nucleophile
Order of Reactivity
- Primary alkyl halides are more reactive toward reactive due to less stearic hindrance.
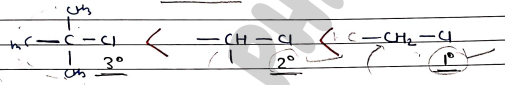
factor affecting reactions
- Substrate Primary substrates are more reactive than tertiary substrates due to less steric hindrance.
- Leaving Group Good leaving groups ($I > Br > Cl$) increase rate.
- Nucleophile Strong nucleophile (OH^-, CN^-$) favors $SN_2 reactions.
- Solvent Polar, aprotic solvents (Acetone) facilitate reactions.
- Steric hindrance Increased steric hindrance decreases rate of reaction of .
VS
| • Two-step mechanism | • One-step mechanism |
| • First order kinetics | • Second order kinetics |
| • Order of reactivity | • Order of reactivity |
| • Unimolecular reactions | • Bimolecular reactions |
| • Weak nucleophiles favor mechanism | • Strong nucleophiles favor mechanism |
STRUCTURE AND USES (Alkyl Halides)
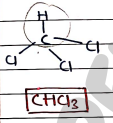
1. Chloroform
- It is a colorless, sweet-smelling organic compounds that is a Trihalomethane.
- It is used as solvent in various industrial and laboratory applications.
- Historically, it was used as general anesthetics.
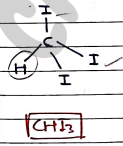
2. Iodoform
- Tri-iodomethane, methyl tri-iodide.
- It is used as disinfectant and antiseptic in medicines.
- Antimicrobial agents.
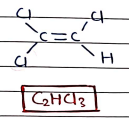
3. Trichloroethylene
- It is used as a solvent for cleaning and degreasing metal parts, equipments, textiles.
- It also act as refrigerant.
- also used as solvent in pharmaceuticals.
ALCOHOLS
These are a class of organic compounds that contain hydroxyl ($-OH$) functional group attached to a carbon atom.
General structure
where R is an alkyl group (carbon chain).
Types
- Primary Alcohols (1^\circ$) $\rightarrow One alkyl group are attached with carbon, which bonded to group.
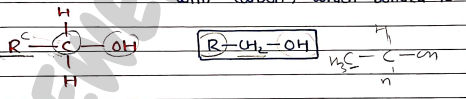
- Secondary Alcohols ($2^\circ$) Two alkyl groups are attached with carbon bonded to group.
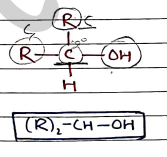
- Tertiary Alcohols (3^\circ$) $\rightarrow Three alkyl groups are attached with carbon bonded to group.
QUALITATIVE TESTS FOR ALCOHOLS
These are those chemical tests which are used to identify the presence or absence of alcohol in a sample. It is of followings:-
- Sodium Metal tests
- Lucas tests
- Ceric Ammonium Nitrate test
- Iodoform tests
- Victor meyer test
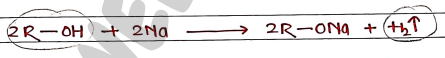
1. Sodium Metal tests When Alcohols reacts with active metals (e.g. Sodium), it liberate hydrogen gas ($H_2$) in the form of effervescence.
2. Lucas tests When Alcohols reacts with Zinc chloride ($ZnCl_2$) and Hydrochloric acid ($HCl$), it produce or formed cloudy layer or precipitate. (Alkyl halides)

- alcohols reacts slowly / no reaction
- alcohols reacts moderately.
- alcohols reacts quickly.
3. Ceric Ammonium Nitrate test When alcohol reacts with ceric ammonium nitrate, it forms red colored alkoxy cerium (IV) compound.
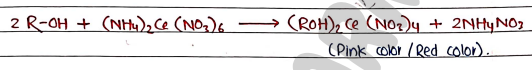
4. Iodoform test When Alcohols warm with sodium hydroxide and iodine, it forms a yellow ppt of iodoform.
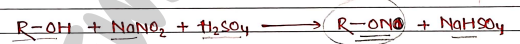
5. Victor Mayer test When Alcohols reacts with sodium nitrite ($NaNO_2$) and sulfuric acid ($H_2SO_4$), it formed a colored solution/precipitate. $R-OH + NaNO_2 + H_2SO_4 \rightarrow R-ONO + NaHSO_4$
- Alcohols Red color.
- Alcohols Blue color
- Alcohols Green color
STRUCTURE AND USES (Alcohols)
1. Glycerol - From Alkyl halides.
- It is used as a solvent for cleaning and degreasing metal parts, equipments, textiles.
- It also act as refrigement.
- also used as solvent in pharmaceuticals.
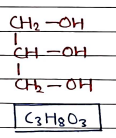
2. Glycerol
- Also known as glycerine or glycerin. It is colorless, odorless and viscous liquid with sweet taste.
- widely used in skin lotions, mouth washes, cough medicines, drug solvents, serum, vaccines and suppositories.
3. Ethyl Alcohol / Ethanol
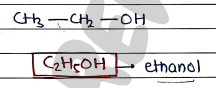
- Used as solvent in pharmaceuticals.
- Used as solvent, preservatives.
- Disinfectant and Antiseptic.
- Also used in production of plastic fibers and other chemicals.
4. Benzyl Alcohol
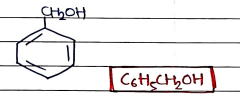
- Used as solvent, preservatives, and flavaring agents.
- Also used in skincare products, soops and lotions.
- Antiseptic, antifungal, anti-bacterial agent.
