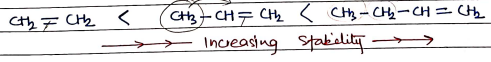Pharmaceutical Organic Chemistry 1 - Unit 2
Syllabus
Alkanes\*, Alkenes* and Conjugated dienes*
SP3 hybridization in alkanes, Halogenation of alkanes, uses of paraffins. Stabilities of alkenes, SP2 hybridization in alkenes E1 and E2 reactions - kinetics, order of reactivity of alkyl halides, rearrangement of carbocations, Saytzeffs orientation and evidences. E1 verses E2 reactions, Factors affecting E1 and E2 reactions. Ozonolysis, electrophilic addition reactions of alkenes, Markownikoff’s orientation, free radical addition reactions of alkenes, Anti Markownikoff’s orientation. Stability of conjugated dienes, Diel-Alder, electrophilic addition, free radical addition reactions of conjugated dienes, allylic rearrangement
Scroll to Download
POC-1ST
UNIT-2
1. ALKANES
- Alkanes are those organic compounds which are composed of hydrogen and carbon only with single bond.
- They are the simplest and most saturated type of hydrocarbons, as they contain and single covalent bond.
General formula
- The general formula for alkanes is where is the number of carbon atom.

Structure
- Alkanes have a linear or branched chain structure, with each carbon atom bonded to four other atoms (hydrogen/carbon).

HALOGENATION OF ALKANES
- It involves the substitution reactions, in which Halogen atom replaces H-atom from the alkane.

- Chlorination, Bromination etc..
Chlorination
- Alkanes react with chlorine in the presence of ultraviolet light to produce alkyl chloride.

- The reaction does not stop at this stage, the remaining three hydrogen atoms of methyl chloride can be successively replaced by chlorine atom.

Mechanism
- The mechanism of chlorination/halogenation take place through the formation of free radical.
- It involves three steps:
- Chain initiation
- Chain propagation
- Chain termination
1. Chain initiation
- In this, chlorine molecules undergoes homolytic fission to give chlorine free radicals.

2. Chain propogation
It further involves two steps:
- a) in this, chlorine free radical attacks methane to produce methyl free radicals and .
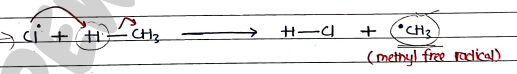
- b) Now, in this, methyl free radical attacks chlorine molecules to give methyl chloride and chlorine free radicals.

$$CH_3^\bullet + Cl_2 \rightarrow CH_3-Cl + Cl^\bullet$$
(Methyl chloride)
3. Chain termination
- Now, in this step two free radicals combine to form a stable molecules, ending the chain reactions...

(methyl chloride)
SP³ HYBRIDIZATION IN ALKANES
Hybridization It is the concept of mixing of atomic orbitals to form a new hybrid orbitals.
- It is the process of combining two or more atomic orbitals of an atom to form equivalent hybrid orbitals.
sp³ hybridization
- It is a type of orbital hybridization in which one orbital and three orbitals of an atom to mix to form four equivalent hybrid orbitals.

- Each hybrid orbital show 25% s-orbital characteristics and 75% p-orbital characteristics.


For Methane ($CH_4$)
- Hybridisation occurs only for central atom.
- Here, Carbon is our central atom.
- It involves several steps:
Steps
- Electronic configuration of central atom.
- Carbon Atomic No 6 (Ground state)
- contains , contains , contains
- At an excited state, always in outermost (orbitals)
- contains , contains
- hybridisation
- overlapping of orbitals
Geometry
- Alkanes show tetrahedral arrangement.
- Hydrogens have oval shaped orbitals.
- The angle b/w bond is 109.5°.
For Ethane
- Both Carbon have hybridisation, so, ethane hybridisation.
PROPERTIES OF ALKANES
- Alkanes are insoluble in water and soluble in organic solvents.
- Alkanes from are colorless gases, to are colorless liquids and higher molecules are solid..
- They are non-polar compounds.
- Boiling point and melting point are increases with increases in molecular weights.
PARAFFINS
- Paraffins are just another name of alkanes.
- They are types of saturated hydrocarbon with a straight or a branched chain structure.
Uses of Paraffins
- They are used as fuels, such as methane (natural gas), propane and butane etc.
- They are used as lubricants in various industrial applications.
- Also used as solvents in the production of coating, paints & adhesives.
- Liquid paraffin, highly refined liquid mineral oil, so it is used as excipients in pharmaceutical products and used in cosmetics and personal care products, such as lotions, creams & Ointments.
- used to clean dry skin, constipation and eczema (skin disease).
2. ALKENES
- These are those unsaturated hydrocarbons, which contain one or more Carbon-Carbon double bond .
- Also known as Olefins.
General formula
- where n is the no. of carbon atoms. (min. 2 carbon required).

Examples:
- (Ethene):
- (Propene):
- Butene etc..
STABILITIES OF ALKENES
- Alkenes are unsaturated hydrocarbons with a Carbon-Carbon double bond .
- They contains double bond, which make alkenes more reactive than alkanes but less stable.
- The stability of alkene depends on:-
Substituents Alkyl group (e.g. methyl, ethyl) stablize the double bond by donating electrons. work as donating R group.
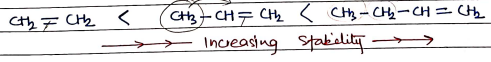
(Increasing Stability)
Trans form are more stable than Cis-form due to less steric hindrance.

- Trans-form: Same groups are at opposite side.
- Cis-form: Same groups are at same side.
HYBRIDIZATION IN ALKENES
- hybridization is a type of orbital hybridization, where one ($s$) orbital and two ($p$) orbitals mix to form three equivalent hybrid orbitals.
- In alkenes, the carbon atom involved in the double bond undergoes hybridization.

e.g. Ethene ($CH_2=CH_2$)
- for central atom:
- Ground State:
- Excited state:
- Now, in hybridised state: hybridization ($s$ and two orbitals mix, leaving one unhybridized orbital).
- Bond angle is 120°.
E1 AND E2 REACTIONS
- and are part of Elimination reactions.
- These are those reactions which involve the removal of a leaving group from a substrate, resulting in the formation of a double bond (ethene / alkenes).
- These are those reactions, which are used in the preparation of alkenes by using dehydrohalogenation of alkyl halides. (de hydro + halogenation).

- E.g. (ethene)
- These are of two types:
- reactions
- reactions
E1 REACTIONS
- It stands for Unimolecular Elimination Reactions.
- It is Unimolecular, so it follow first-order kinetics i.e. it is depends on the concentration of substrate (reactant only).
- It is a type of reaction, which include two-step mechanism.
- Tertiary alkyl halides are more reactive towards this reaction. Order of reactivity
Mechanism
- It is two step mechanism, in which first step is rate-determining step:-

(tertiary butyl bromide 2-methyl propene + Hydrogen bromide)
- Step-1 It involves the formation of carbocation (intermediate). It is rate-determining step. (slow step)

(Alkyl halides Carbocation)
- Step-2 It involves the deprotonation. (fast step)
(Note: Since, the first step is slow, and rate-determing, the overall rate of reaction depends only on the reactant/substrate.)
Kinetics
- It follow 1st order kinetics, because in reactions the rate of reactions depends on the concentration of the substrate/reactant. where Rate of reaction, Rate constant, concentration of alkyl halide
Order of Reactivity
- It follow . In reactions, order of reactivity depends on the stability of Carbocation. (3° have more substituent more stable).
E2 REACTIONS
- represents (Bimolecular) Elimination Reactions.
- It is bimolecular, so it follow second order kinetics i.e. it depends on the concentration of both (reactant & reagent).
- It involves one-step mechanism.
- alkyl halides are more reactive toward this reactions. Order of Reactivity
Mechanism

Reaction (ethyl bromide + potassium bromide ethene)
It completed in one step, which include transition state.

- In transition state (the rate-determining step), both ethyl bromide and hydroxide ion involved.
Kinetics
- It follow second order kinetics, rate of reaction depends on both reactant and reagent. $$R = K [alkyl halides][base]$$
Order of Reactivity
- 1° alkyl halides are more reactive toward reactions, due to less stearic hindrance. can easily attack. .
E1 VERSUS E2
| E1 Reactions | E2 Reactions |
|---|---|
| • Unimolecular | • Bimolecular |
| • involve two steps | • involve one step only |
| • formation of carbocation | • formation of transition state |
| • | • |
| • weak base required | • strong base required |
| • Highly polar solvents | • low polar solvents |
| • Rearrangement of carbocation possible | • No rearrangement occurs |
FACTOR AFFECTING E1 & E2 REACTIONS
- Substrate For : (due to stability of carbocations). For : (due to low stearic hindress).
- Leaving group Good leaving group (e.g. Cl, Br, I) facilitate reacting.
- Solvent For : polar protic solvents (e.g. water). For : polar aprotic solvents.
- Nature of Base For : weak base. For : strong base.
- Temperature high temp facilitate both & reactions.
RE-ARRANGEMENT OF CARBOCATIONS
- It is the shifting/migration of Hydrogen/methyl group from one Carbon to another to form a more stable carbocations.
- It is of two types:-
- Hydride shift A hydrogen atom migrates from one C to another.

more stable. 2. Methyl shift A alkyl group ($-CH_3$) migrates from one C to another.

$CH_3-C(CH_3)(CH_3)-CH^+-CH_3 \rightarrow CH_3-C^+(CH_3)-CH(CH_3)-CH_3$ (more stable tertiary).
SAYTZEFF'S RULE (Zaitsev's Rule)
- It occurs in elimination reactions.
- When alkyl halides have two or more $\beta$-carbon present, it used in this case to identify the major and minor product.

- (But-2-ene/2-Butene) 80%
- (But-1-ene/1-Butene) 20%
- Acc to this rule, that product will be the major product, which contain more no. of substituents at double bond carbon.
- So, in above example, But-2-ene will be the major product.
- Evidences It helps us understand and predict the outcomes of elimination reactions. predict the major product formed.
ELECTROPHILIC ADDITION REACTIONS OF ALKENES
- These are those reaction, in which electrophile ($E^+$) added to the double carbon by breaking down double bond.

- It involves the formation of alkanes from alkenes.

(ethene + Hydrogen bromide Bromo ethane)
Mechanism
- It involves three steps:

- Formation of electrophile ($E^+$)
- Attack of electrophile on reactant (ethene) (formation of carbocation)
- Now, addition of Nucleophile ($Nu^-$) on carbocation (Bromoethane)
Now, Alkenes are of two types:-
- Symmetrical alkenes: when alkyl/hydrogen are same on both sides of double bond.
- Unsymmetrical alkenes: when alkyl/hydrogen are unequal on both sides of double bond.
Now, Markovnikoff and Anti-markovnikoff rules are applied on unsymmetrical alkenes.
MARKOVNIKOFF'S RULE * This rule is given by russian chemist markovnikov.
- Acc. to this rule: In an electrophilic addition reactions, Electrophile ($E^+$) goes to that double bond carbon, where more number of hydrogen are present.
- OR Nucleophile ($Nu^-$) goes to that double bond carbon, where less number of hydrogen are present.

(2-bromopropane)
ANTI-MARKOVNIKOFF'S RULE
- Also known as Kharasch effect or Peroxide effects ($H_2O_2$).
- It occurs in assymetrical alkene.
- According to this rule: Hydrogen atom added to those double bond carbon, which contain less number of hydrogen/non-polar nature.

(1-bromopropane)
- It involves the free radical addition mechanism.
Mechanism
- It involve three steps:-

- Chain initiation It occurs in reagent and catalyst. (Bromine free radical)
- Chain propogation Now, Bromine free radical attack reactant. $$Br^\bullet + CH_3-CH=CH_2 \rightarrow CH_3-C^\bullet H-CH_2-Br$$
- Chain termination free radicals reacts with each other. $$CH_3-C^\bullet H-CH_2-Br + H-Br \rightarrow CH_3-CH_2-CH_2-Br + Br^\bullet$$ (1-bromopropane) $$Br^\bullet + Br^\bullet \rightarrow Br_2$$
OZONOLYSIS
- It is an organic reaction, where an alkene is cleaved by ozone ($O_3$) to form two carbonyl compounds (Aldehydes or ketones).

(ethene 2 formaldehyde)
Mechanism
- It involves two steps:-
- Formation of ozonide Ethene reacts with ozone to form an ozonide intermediate.

$$CH_2=CH_2 + O_3 \rightarrow \text{Ozonide (cyclic structure with 3 oxygen atoms)}$$
- Reduction Now ozonide reduced to form two carbonyl compounds.

$$\text{Ozonide} \xrightarrow{Zn/H_2O} 2(CH_2=O) + ZnO$$
(formaldehyde)
CONJUGATED DIENES
- These are those type of organic compounds that contains two or more Carbon-carbon double bonds ($C=C$) separated by a single bond in alkenes.

e.g.
- General structure where .
- they contains, alternate double bonds. e.g. (1,3-Butadiene)
CHEMICAL REACTIONS
It undergoes following chemical reactions:-
- Electrophilic addition reactions
- Free radical reactions
- Diels-alder reactions
1. ELECTROPHILIC ADDITION REACTIONS
- These are those reaction in which electrophile ($E^+$) added to the carbon atom and breakage of one double bond ($\pi$-bond) take places.
- In this reaction, 1,3-Butadienes are react with halogens ($Br_2$) in the presence of an inert solvent ($CCl_4$) to give a mixture of two di-bromo compounds.

- (1,2-addition, 3,4-dibromo-1-butene)
- (1,4-addition, 1,4-dibromo-2-butene)
Mechanism

- Formation of carbocation
- Combination of bromide with carbocation
- 1,2-addition product
- 1,4-addition product
2. FREE RADICAL ADDITION REACTIONS
- Conjugated dienes can undergoes free radical reactions, which involves the addition of free-radical to the double bond carbon.
- It involves the addition of 1,3-butadiene to bromotrichloromethane ($BrCCl_3$) in the presence of an peroxide to yield 1,2- and 1,4- addition products.

- (1,2-addition)
- (1,4-addition)
Mechanism
- It involves three steps:

- Chain initiation formation of free radicals.
- Chain propogation addition of free radicals to diene. $$CH_2=CH-CH=CH_2 + \cdot CCl_3 \rightarrow CH_2(CCl_3)-C^\bullet H-CH=CH_2 \leftrightarrow CH_2(CCl_3)-CH=CH-C^\bullet H_2$$
- formation of 1,2- and 1,4- addition products by reaction with .

3. DIELS - ALDER REACTIONS * It involves the treatment of 1,3-Butadiene (conjugated dienes) with an alkene/alkyne to form cyclohexane.
- It does not required any catalyst.
- Diene + Dienophile (alkene/alkyne) Adduct (formed product).
- Given by Paul Hermann Diels and Kurt Alder and get nobel prize.

Mechanism
- It involves single step process, in which dienophile interact with diene and formed adduct.

- In this, six-membered ring compounds are formed in large amount.
ALLYLIC REARRANGEMENT
- It is a type of organic reaction where an allylic group (a functional group attached to carbon atom adjacent to a double bond) shift to a nearby position, results in more stable compounds.

e.g.