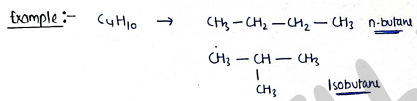Pharmaceutical Organic Chemistry 1 - Unit 1
Syllabus
Classification, nomenclature and isomerism
Classification of Organic Compounds Common and IUPAC systems of nomenclature of organic compounds (up to 10 Carbons open chain and carbocyclic compounds) Structural isomerisms in organic compounds
Scroll to Download
POC-1ST
UNIT 1ST
CLASSIFICATION, NOMENCLATURE, AND ISOMERISM
SYLLABUS
- Classification of organic compounds
- Common and IUPAC system of Nomenclature of organic chemistry
- Structural isomerism in organic compounds.
Organic Chemistry
It is the branch of science, which deal with the study of molecules which is made up of Carbon [C].
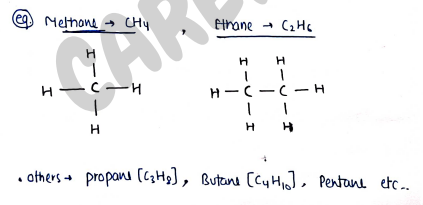
- In mostly, carbon is attached with Hydrogen. So, it is also known as Hydrocarbons. e.g. (Methane), (ethane) etc..
CLASSIFICATION OF ORGANIC COMPOUNDS
Organic compounds
These are those compounds which is made up of carbons.
Eg. Methane , Ethane etc..
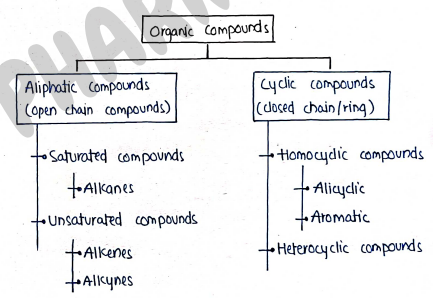
Aliphatic compounds
Also known as Open chain structure.
- These are those compounds in which carbon atom are attached together in the form of straight branched chain, not in cyclic/closed structure.

i) Saturated compounds : These are those organic compounds in which carbon attached with carbon with single bond, such as "$C-C$". Eg. Alkanes , (Methane) (Ethane) etc..
- Alkanes : These are saturated hydrocarbons with general formula . They contains only carbon-carbon and carbon-hydrogen single bond in their molecules.
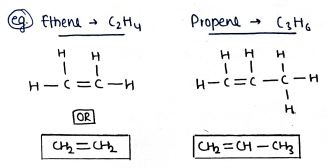
ii) Unsaturated compounds :- These are those organic compounds in which carbon attached to carbon with atleast one double or triple bond. e.g. Alkenes, Alkynes.
- Alkenes : Alkenes are the unsaturated hydrocarbons with general formula . They contain atleast one carbon to carbon double bond in their molecules.
- Alkynes : Alkynes are the unsaturated hydrocarbons with general formula . They contain atleast one carbon to carbon triple bond in their molecules.

Cyclic Compounds
These are those organic compounds which contain cyclic structure or closed rings like structure.
e.g. Cyclopropane
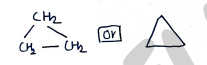
i) Homocyclic compounds : These are those cyclic organic compounds in which the ring forming atoms are only carbon.
They also termed as carbocyclic compounds.
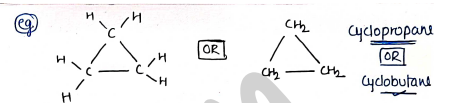
Alicyclic : These are those in which carbon atoms are attached in closed structure but they do not have aromatic character. It may be saturated or unsaturated.
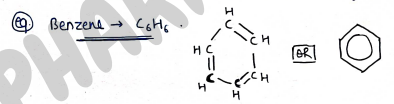
- Aromatic : These are those organic compounds which follow huckle's rule of aromaticity.
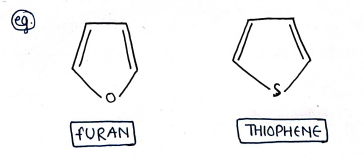
ii) Heterocyclic compounds : These are those cyclic organic compounds in which atleast one heteroatom (i.e. atoms other than carbon e.g. N, O, S) is present as one of the ring forming atom.
NOMENCLATURE OF ORGANIC COMPOUNDS
- There are several organic compounds present in our environment.
- It is necessary to named these compounds for their unique identification.
- There are mainly two nomenclature system used for naming organic compounds :-
- Trivial or common system
- IUPAC System
1. Trivial or common system
In this, compound should be named on the basis of various factors such as source, name of discoverer, place, structure etc..
- Wood spirit (obtained by destructive distillation of wood.)
- Oxalic acid (Obtained from oxalis plant.)
- Glucose, Glycerol sweet in taste (properties) etc..
2. IUPAC System
IUPAC stands for "International Union of Pure and Applied chemistry"
- It is a standard and systematic method of naming organic compound, based on their structure.
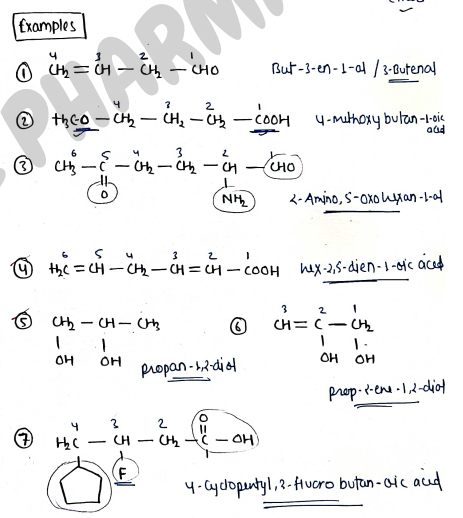
- A compound is named using IUPAC by following - firstly identifying the parent chain of hydrocarbon then functional group, prefixes, suffixes--
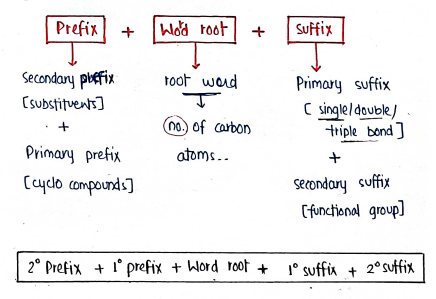
- The word root is the name of organic compound is the no. of carbon atoms....
| No. of C atom | Word root | Example Formula | Example Name |
|---|---|---|---|
| 1 C | Meth | Methane | |
| 2 C | Eth | Ethane | |
| 3 C | Prop | Propane | |
| 4 C | But | Butane | |
| 5 C | Pent | Pentane | |
| 6 C | Hex | Hexane | |
| 7 C | Hept | Heptane | |
| 8 C | Oct | Octane | |
| 9 C | Non | Nonane | |
| 10 C | Dec | Decane | |
| 20 C | Icos | Icosane | |
| 30 C | Triacont | Triacontane |
- Alkyl (R-) Group : It is obtained by removing one hydrogen atom.
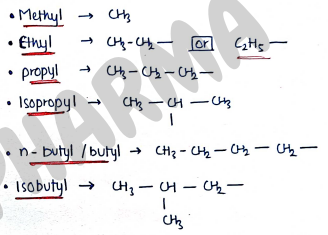
- Bonds :
- Single (-) bond ane
- Double (=) bond ene
- Triple (\equiv$) bond $\rightarrow yne
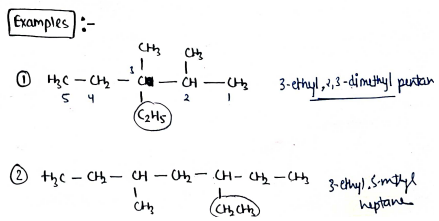
GENERAL RULES FOR NOMENCLATURE
- select the longest chain, and starts the numbering from that side, where no. of substituents are more.
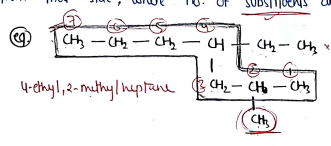
- If the parent chain has two or more substituents at equivalent position, then give priority through alphabetically.
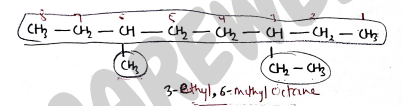
The no. of position is written by using commas and the appropriate prefix - di, tri, tetra etc..
If the substituent also carries a branched chain [complex substituents], then the naming of that substituent done separately and written in bracket.
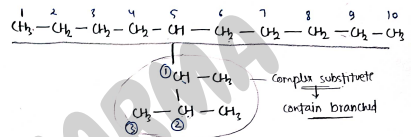
- The unsubstituted radicals / branched name can also be using their trival names.
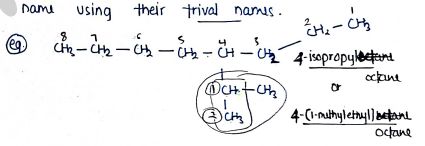

FOR ALKENES & ALKYNES
- Alkenes are double bonded compounds, and for these compounds suffix "-ane" is replaced to "-ene".
- Alkynes are triple bonded compounded and for these compounds suffix "-ane" is replaced to "-yne".
- If parent chain contain double/triple bond, then numbering start from double/triple bond side. If both, then give priority to double bond. ($=$ > $\equiv$).
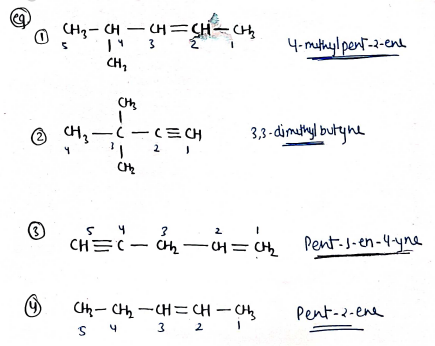
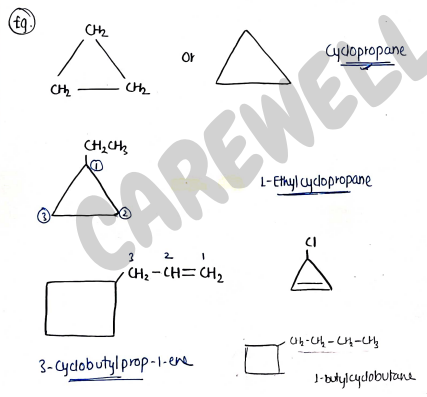
FOR CYCLIC COMPOUNDS
- for cyclic compounds, add prefix - cyclo before the main chain name.
- Give priority of large no. of carbon, if same the give priority to ring.
- If double/triple bond present, then give priority to them.
FOR FUNCTIONAL GROUP
- Always give first priority to functional groups.
- functional group is written by adding secondary suffix after 1° suffix, 'e' terminal of primary suffix is removed before 2° suffix if its name begins with a, i, o, u or y, e.
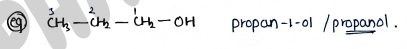
- If compound contain more than one functional group that highest priority group will be choose and other group work as substitute.
- Halogens [Cl, Br, I] also choose as substituent.
Some commonly used functional group
| Functional group | Prefix | Suffix | |
|---|---|---|---|
| 1 | Carboxylic acid ($-COOH$) | Carboxy | oic acid |
| 2 | Aldehyde ($-CHO$) | formyl | al |
| 3 | Alcohol ($-OH$) | Hydroxy | -ol |
| 4 | Ketone ($=O$) | Oxo | -one |
| 5 | Ether ($R-O-R'$) | Alkoxy | - |
| 6 | Ester ($R-CO-OR'$) | Alkoxy carbonyl | oate (Alkyl alkanoate) |
| 7 | Cyanide / Nitrile ($-CN$) | Cyano | Nitrile |
| 8 | Amide ($-CONH_2$) | Carbamoyl | -amide |
| 9 | Amine ($-NH_2$) | Amino | Amine |
| 10 | Thiol ($-SH$) | mercapto | thiol |
| 11 | Sulphonic acid ($-SO_3H$) | -Sulphonic acid |
- Priority for functional group :
Salts > COOH > Anhydride > Ester > Acid chloride > Amide > Nitrile > Aldehydes > ketone > Alcohol > Amine > Ether > alkene/alkyne.
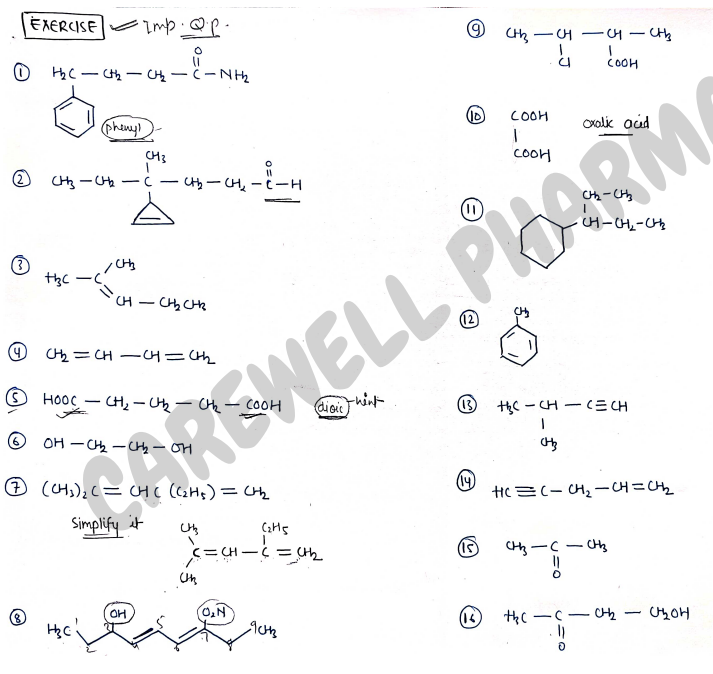
EXERCISE
ISOMERISM
Isomers are those organic compound that have similar molecular formula but different structure or different in their physical or chemical properties.
This phenomena is known as isomerism.
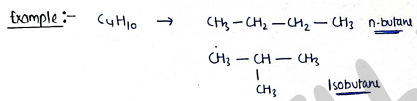
Classification of Isomerism
- It is majorly divided into two main parts :-
- Structural isomerism
- Stereoisomerism
1. STRUCTURAL ISOMERISM
- Those compounds which have same molecular formula but they have different structural formula are known as structural isomers and this phenomena is known as structural isomerism.
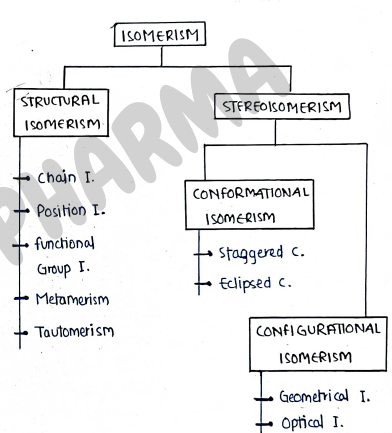
- It is further sub-divided into five types
- Chain isomerism
- Position isomerism
- Functional isomerism
- Metamerism
- Tautomerism
I. Chain isomerism:- It contains those isomers which have same molecular formula but different in their chain structure.
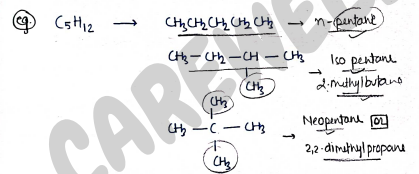
e.g.
- n-pentane
- Isopentane (2-methylbutane)
- Neopentane (2,2-dimethyl propane)
II. Position isomerism:- It contains those isomers which have same molecular formula but different in position of functional group.
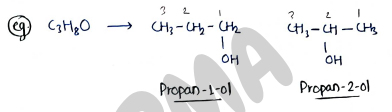
e.g.
- (Propan-1-ol)
- (Propan-2-ol)
III. Functional isomerism:- It contains those isomers which have same molecular formula but different functional group.
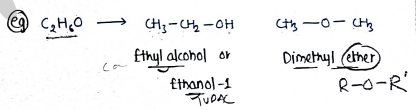
e.g.
- (Ethyl alcohol or Ethanol)
- (Dimethyl ether)
IV. Metamerism:- It contains those isomers, which have same molecular formula but unequal distribution of carbon atoms/alkyl groups attached to a functional groups.
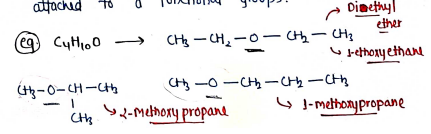
e.g.
- (Diethyl ether / ethoxyethane)
- (1-methoxypropane)
- (2-Methoxy propane)
V. Tautomerism:- It contains those isomers/molecules which have same molecular formula but contain two or more inter-convertible structures.
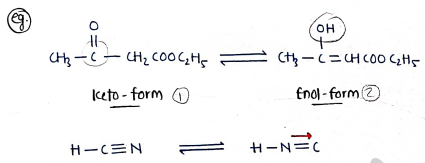
e.g.
- (keto-form Enol-form)
2. STEREOISOMERISM
- Those compounds which have same molecular formula, but different arrangement of atom/molecules in a 3D space, are known as stereo isomers.
- This phenomena is known as stereoisomerism.
- They have different configuration i.e. 3D arrangement of atoms.
I. Geometrical isomerism:- Also known as Cis-trans isomerism.
- It contains those isomers which have same molecular formula but differ in the spatial arrangement of atoms/group about the double bond.
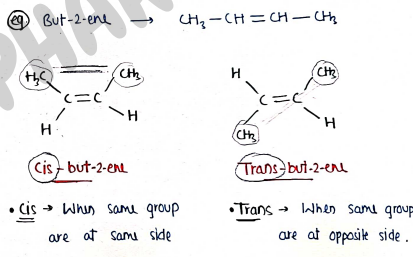
e.g. But-2-ene ($CH_3-CH=CH-CH_3$)
Cis-but-2-ene: When same group are at same side. * Trans-but-2-ene: When same group are at opposite side. II. Optical isomerism:- It is the type of stereo-isomerism, which contains those isomers which have same molecular & structural formula but differ in their behaviour towards light ie plane polarised light.
Those compounds which rotates the plane polarised light (ppl) are known as optically active compounds.
Those not rotate are optically inactive compounds. e.g. Lactic acid [$C_3H_6O_3$]
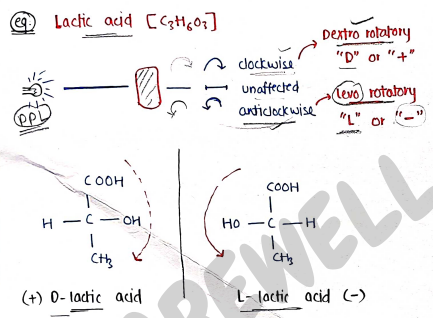
- (+) D-lactic acid: Dextro rotatory "D" or "+" (clockwise)
- L-lactic acid (-): Levo rotatory "L" or "-" (anticlockwise)
Enantiomers:- Molecules that are mirror image of each other and non-superimposable.
Diastereomers:- non-superimposable & non-mirror images.
III. Conformational Isomerism:- It contains those isomers which have same molecular formula but differ in the spatial arrangement about single bond.
- It occurs due to rotation around a single bond formed conformational isomers/conformers.
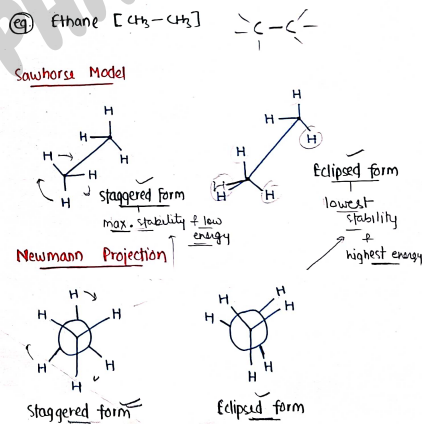
e.g. Ethane [$CH_3-CH_3$] * Sawhorse Model & Newmann Projection:
- Staggered form: max. stability & low energy.
- Eclipsed form: lowest stability & highest energy.