Pharmaceutical Inorganic Chemistry - Unit 4
Syllabus
Miscellaneous compounds
Expectorants: Potassium iodide, Ammonium chloride*.
Emetics: Copper sulphate\*, Sodium potassium tartarate
Haematinics: Ferrous sulphate\*, Ferrous gluconate
Poison and Antidote: Sodium thiosulphate\*, Activated charcoal, Sodium nitrite333
Astringents: Zinc Sulphate, Potash Alum
Scroll to Download
MISCELLANEOUS COMPOUNDS
UNIT-4
SYLLABUS
- Expectorants - Potassium Iodide, Ammonium chloride*
- Emetics - Copper Sulphate, sodium potassium tartarate
- Haematinics - ferrous sulphate, ferrous gluconate
- Poison & Antidote - Sodium nitrite, Sodium thiosulphate, Activated charcoal
- Astringents - Zinc sulphate, Potash Alum.
EXPECTORANTS
- These are those agents/drugs which are used to expel sputum (mucus + cell debris + foreign particles) from the respiratory tract and helps in the treatment of cough.
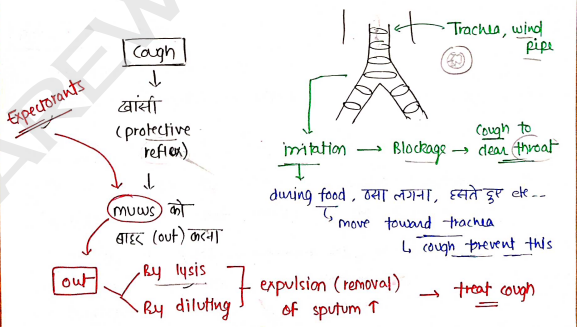
- These agents are used to clear thick mucus from the respiratory tract including lungs, bronchi and trachea, and providing relief from respiratory tract disorders.
Classification : Based on the mechanism of action, it is mainly
classified into two types:
Secretion Enhancers : They mainly give their function by increasing the bronchial secretion of mucus, which dilute the sputum and helps in its removal.
- e.g. Guaifenesin most common, Potassium Iodide and Ammonium chloride*.
Mucolytics : They mainly give their action by breaking the mucus and reduce its viscosity with the action of enzymes.
- e.g. Bromhexine, Ambroxol etc..
POTASSIUM IODIDE
- Molecular formula:
- Molecular weight:
Method of Preparation :
- It is prepared by treating potassium bicarbonate with hydrogen iodide, which results in the formation of .

- It is also prepared by treating hot aqueous solution of potassium hydroxide with iodine.
Properties :
- It is an odourless, transparent with saline bitter taste.
- It is soluble in alcohol, water and glycerine.
- It is hygroscopic in nature.
Uses :
- It is used as an expectorants.
- It is also act as a source of potassium and iodine.
- also used as reagent in pharmacy.
AMMONIUM CHLORIDE
- Molecular formula:
- Molecular weight:
Method of preparation :
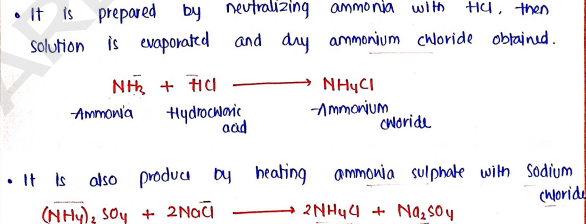
Properties :
- It is a colorless/white, crystalline / course powder.
- It is odourless with saline taste.
- It is freely soluble in water and glycerine, but sparingly soluble in alcohol.
- It is slighty hygroscopic.
Reactions :
Assay : Currently, acid-base titration is preferred for the assay of ammonium chloride.
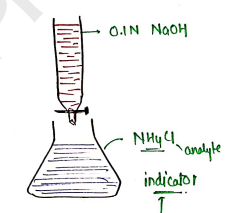
- Weight of & dissolve in of .
- Now, add a mixture of previously neutralized formaldehyde solution ($5 \text{ ml}$) & stand by for two minutes.
- Now, titrated against solution, using phenolphthalein as an indicator.
- End point is the appearance of pale pink color.
- Each ml of of .
Uses :
- It is used as expectorants.
- It is also used as acidifiers to maintain acid-base balance.
- It also act as diuretics.
Storage : It is stored in tightly closed container.
EMETICS
- These are those drugs which helps in emptying the stomach by inducing vomiting.
- These are those drugs which induce vomiting.
- Vomiting is the forceful expulsion (out) of gastric content from the body through mouth.
- They are mainly used in the case of:
- poisoning
- overdose of any drugs
- Adverse effects
- wrong medications. etc..
Mechanism :
- Emetics stimulates the chemoreceptor trigger zone (CTZ) present in medulla oblongata, which stimulates vomiting centre (VC) and induce vomiting. e.g. morphine, digitalis etc..
- By irritating the GIT, which increase secretion & movement. e.g. zinc sulphate, Copper sulphate, sodium chloride etc.
Characteristics :
- It should be non-toxic.
- Non interactive with other drugs.
- non-irritant.
- short onset of action.
Examples : Copper sulphate, sodium potassium tartarate
1. COPPER SULPHATE *
It is present in hydroform and also known as blue vitriol.
- Molecular formula :
- Molecular Weight :
Method of preparation :
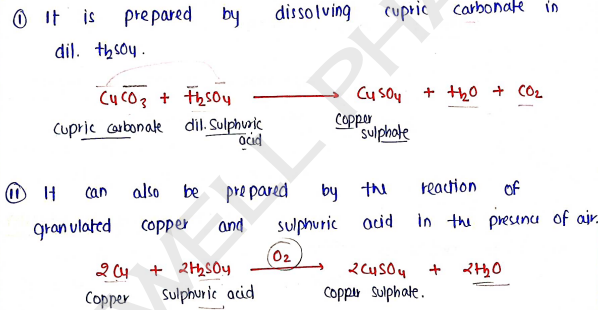
Properties :
- It occurs as deep blue color crystals or blue powders.
- It is odourless.
- It is soluble in water and insoluble in alcohol.
Reactions : Copper sulphate and potassium iodide reacts together to form unstable cupric iodide. $$2CuSO_4 + 4KI \rightarrow 2CuI_2 + 2K_2SO_4$$
ASSAY :
- A suitable amount of copper sulphate is weighed and dissolved in water.
- and acetic acid are added in this solution.
- Now, iodine ($I_2$) is obtained, which is then titrated with standard sodium thiosulphate solution, using starch as an indicator.
- Titration continues until the blue color disappears.
Uses :
- It is used as emetics.
- It is also used as astringent and a fungicide.
SODIUM POTASSIUM TARTARATE
It is also known as rochelle salt.
- Molecular formula:
- Molecular weight:
Method of preparation : It can be prepared by neutralising a solution of sodium carbonate with potassium bitartrate.
Properties :
- white/colorless crystalline powder.
- odourless.
- saline taste.
- soluble in water.
- Insoluble in alcohol.
Uses :
- used as emetics.
- also used as saline cathartics.
HAEMATINICS
- These are those drugs or agents which are used to increase the concentration of Haemoglobin (Hb) or iron in blood.
- They are basically used in the treatment of anaemia or Iron-deficiency anaemia.
- These agents are mainly used for the formation of blood.
- Anaemia is a condition of decreased amount of haemoglobin/blood in our body, or mainly loss of RBCs.
Causes :
- Blood loss (acute/chronic). trauma, haemorrhage etc..
- Decrease in RBC formation. Hb, Iron.
Examples : ferrous sulphate, ferrous gluconate
FERROUS SULPHATE
- It is used as haematinic and contain Fe. It is also known as green vitriol.
- Molecular formula:
- Molecular weight:
Preparation :
- It is prepared by treating iron with dil. sulphuric acid, which forms ferrous sulphate and liberated gas.
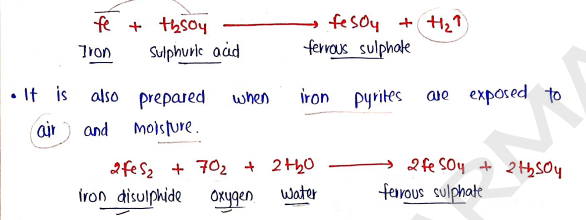
Properties :
- It is pale, bluish green crystals or granules.
- It is odourless and bitter saline taste.
- It is soluble in water and insoluble in alcohol.
Chemical reactions :
- On heating, it decomposed into sulphur dioxide, sulphur trioxide and ferric oxide salts. $$2FeSO_4 \xrightarrow{\Delta} Fe_2O_3 + SO_2 + SO_3$$
Assay :
- It is assay by redox reaction.
- of ferrous sulphate is weighed and dissolved in of dil. . (also used as indicator in this).
- The resultant solution is titrated with .
Uses :
- It is used as haematinics.
- It is also used for treating anaemia (iron - deficiency anaemia).
FERROUS GLUCONATE
- Molecular formula:
- Molecular weight:
- It contains about Fe (iron).
Method of preparation :
- Freshly prepared ferrous carbonate is heated with suitable concentration of gluconic acid in aq. solution to produce ferrous gluconate.
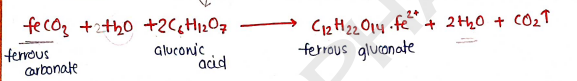
Properties :
- It is yellowish grey powder.
- It have burnt sugar odour.
- It is soluble in cold water and insoluble in alcohol.
Uses :
- It is formulated in tablet dosage form in case of iron deficiency anaemia.
- It is also used in elixir form.
- used as haematinics.
POISON AND ANTIDOTE
- Poison is any substance that cause illness in body or even death after administration in body.
- Poison is administered due to:-
- Intentional when sometime cause to harm himself/herself.
- Unintentional sometime people administered it accidentally.
Symptoms :
- Breathing Problem
- Decreased heart rate
- Dilated pupils
- Vomiting, Diarrhoea
ANTIDOTES
- These are those substances which are used to neutralize the effect of poison.
- These substances are mainly used in the treatment of poisoning.
Classification/Types :
On the basis of mechanism of action, classified into three categories:
- Mechanical Antidotes :
- These are those antidotes which prevent the poison from getting absorbed into the body.
- e.g. activated charcoal.
- Physiological Antidotes :
- These are those antidotes which produce the opposite/reverse effect of poison.
- e.g. Sodium Nitrite.
- Chemical Antidotes :
- These are those antidote which alter/change the chemical nature of poison, which convert the poison into inactive or harmless compound.
- e.g. Sodium thiosulphate.
MECHANISM :
e.g. Cyanide poisoning.
- Normally, during cellular respiration Cells take from blood with the help of enzyme Cytochrome oxidase.
- with the help of Cells generate ATP, Energy.
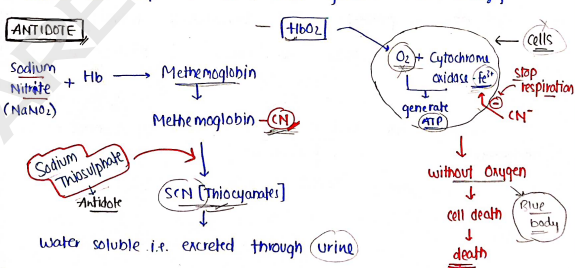
SODIUM THIOSULPHATE
It is also known as sodium hyposulphate. It contains not less than and not more than .
- Molecular formula:
- Molecular Weight:
Method of preparation :
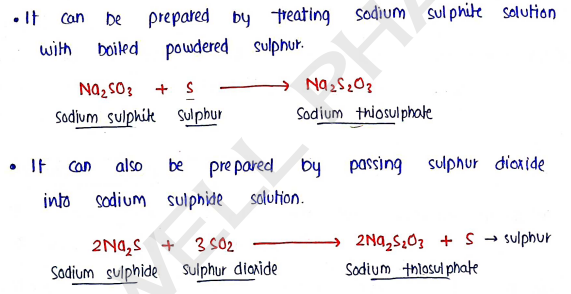
Properties :
- It is colorless, transparent crystals having a bitter salty taste.
- It is soluble in water and insoluble in alcohol.
Chemical reaction :
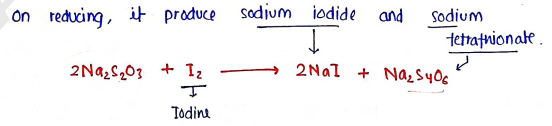
Assay :
- It is assayed by redox titration.
- Take about of dried sodium thiosulphate.
- Dissolved it in of water.
- The resultant solution is titrated with iodine solution.
- using starch as an indicator.
- titration is continues until blue color of solution disappears.
- Each ml of of .
Uses :
- Used in the treatment of cyanide poisoning with sodium nitrite.
- also used as antioxidants, topical antifungal agents.
- also used to treat skin infections.
ACTIVATED CHARCOAL
- It is the residue obtained from the destructive distillation of various organic material.
- It is prepared by using various organic wastes such as sucrose, lactose, rice, starch, coconut etc.
- They give their work by using its absorptive power.
Properties :
- It is fine, black, Odourless and tasteless powder.
- It is insoluble in water and other Organic solvents.
Medicinal uses :
- It is used as an emergency antidote in many forms of poisoning.
- It will adsorb alkaloids, , , , , , and etc.
- It is also used in filters of gas masks.
ASTRINGENTS
- These are those substances which cause protein precipitation.
- These agents are applied topically on damaged skin, mouth and mucous membrane of gastrointestinal tract which forms a protective layer.
Mechanism :
- Astringents forms the protective layer which:-
- protects against bacteria and infections.
- prevents capillary leakage when applied to bleeding areas.
- reduces local oedema, exudation, inflammation and mucus secretion.
- They also reduces the cell permeability because of their capability of precipitating proteins.
Examples : Zinc sulphate, Potash Alum etc..
ZINC SULPHATE
It is also known as white vitriol.
- Molecular Formula:
- Mol. Weight:
Preparation :
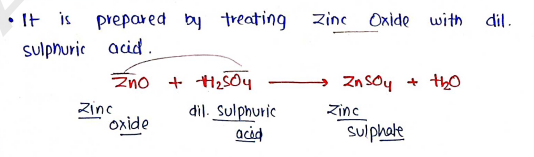

Properties :
- It is colorless, transparent or crystalline powder.
- It is odourless & metallic taste.
- soluble in water & glycerine, insoluble in alcohol.
Uses :
- used as astringents.
- also used as an antiseptic.
- also used for treating acne, dandruff.
- orally administered for healing wounds.
POTASH ALUM
It is also known as potassium aluminium sulphate.
- Molecular formula:
- Molecular weight:
Preparation :
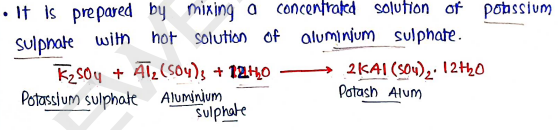
Properties :
- It exists as colorless, transparent or granular crystals.
- sweet astringent taste.
- soluble in water and insoluble in alcohol.
Medicinal uses :
- used topically as astringents.
- also used as antiseptic.
- also used as pharmaceutical aid.
