Pharmaceutical Inorganic Chemistry - Unit 3
Syllabus
Gastrointestinal agents
Acidifiers: Ammonium chloride* and Dil. HCl
Antacid: Ideal properties of antacids, combinations of antacids, Sodium Bicarbonate\*, Aluminum hydroxide gel, Magnesium hydroxide mixture
Cathartics: Magnesium sulphate, Sodium orthophosphate, Kaolin and Bentonite
Antimicrobials: Mechanism, classification, Potassiu permanganate, Boric acid, Hydrogen peroxide\*, Chlorinated lime\*, Iodine and its preparations
Scroll to Download
GASTROINTESTINAL AGENTS
Unit-3
SYLLABUS :
Acidifiers : Ammonium Chloride and Dil. HCl
Antacid : Ideal properties of antacids, combinations of antacids, Sodium Bicarbonate, Aluminium hydroxide gel, magnesium hydroxide mixture
Cathartics : Magnesium sulphate, sodium orthophosphate, kaolin and Bentonite
Antimicrobials : Mechanism, classification, potassium permanganate, Boric acid, Hydrogen peroxide, chlorinated lime, Iodine & its Preparations.
Gastrointestinal Agents
These are those agents or drugs which are used to treat gastrointestinal disorders or disease.
- It involves various inorganic and organic drugs.
Examples : Acidifiers (dil. HCl) and Antacids (sodium bicarbonate) etc..
- Gastrointestinal disorder/disease are those which occurs in gastrointestinal tract.
Gastrointestinal Tract :
- The GI tract is the important system of our body, which consist of Oral cavity, pharynx, oesophagus, stomach, small intestine and anal canal.
- It is the pathway of digestive system.
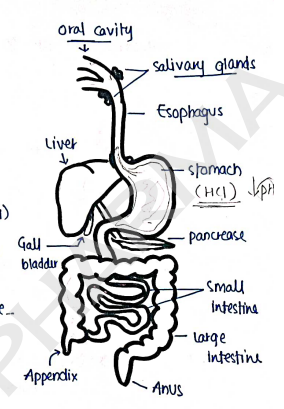
- The main function of GI tract is to digest and absorb nutrients from food, and to excrete waste products of digestion.
- Stomach is the main organ for digestion, because it produce and contain acid i.e. Hydrochloric acid ($HCl$) which play a major role in digestion, but it is very dangerous.
- Excess of HCl Hyperacidity, ulcer etc.
- Deficiency of HCl Achlorhydria or Hypochlorhydria (decrease digestion).
- Intestines involves in absorption and movement of food and at last secretes waste (stool) through anus.
- Low movement constipation (treated by cathartics).
- Microbes can affect any part of GI tract, which is prevented by antimicrobial agents.
ACIDIFIERS
Acidifiers or agents (inorganic substances) or drugs used to increase the metabolic acidosis i.e. to increase the acidity or gastric acid in GI tract (stomach).
- These agents are mainly work by two way, either by decreasing the pH of stomach or by increasing the level of acid (producing acid).
- Also known as Acidifying agents.
- These are used in the treatment of Achlorhydria or Hypochlorhydria.
Examples : Ammonium Chloride and dil. HCl.
Classification
Gastric acidifiers : These are used to treat patients suffering from achlorhydria or hypochlorhydria and helps to restore acidity of stomach.
Urinary acidifiers : Used to control the pH of urine to treat urinary tract infections.
Systemic acidifiers : Used to control the pH of our body by neutralise the alkaline body fluids.
1. Ammonium Chloride
- Molecular formula:
- Molecular weight:
Method of Preparation : It is obtained when ammonia gas is reacts with hydrochloric acid, which further dried by evaporation.

- It can also prepared by the reaction of ammonia gas liquors with lime, liberate ammonia which further reacts with HCl to form . (same as 1st).
Properties :
- It is white crystalline / course powder.
- It is slightly hygroscopic, odourless inorganic salt with a cooling saline taste.
- It is freely soluble in water and glycerin but sparingly soluble in alcohol.
Chemical reaction :
- Ammonium chloride on hydrolysis give ammonium hydroxide.
- On decomposition,
ammonia gas ($NH_3$) + Hydrochloric acid ($HCl$).
Assay :
- Its assay is carried by acid-base titration.
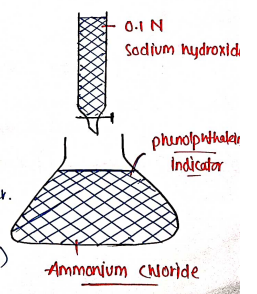
- Take of ammonium chloride in of distilled water.
- Add a mixture of of neutralised formaldehyde solution and dist. water.
- Leave it for 2 minutes, then titrate slowly with sodium hydroxide ($NaOH$) using phenolphthalein as Indicator.
- of of .
Uses :
- Used as acidifier to treat achlorhydria.
- Used as fertilizers and also used as expectorants in cough preparations.
2. Dil. Hydrochloric Acid
- Molecular formula: Dil. HCl
- Molecular weight:
It is a solution of hydrogen chloride in water. ($10% \text{ w/w}$ of HCl)
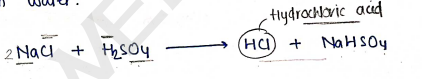
Method of Preparation :
It is prepared by reacting sulphuric acid ($H_2SO_4$) with Sodium chloride ($NaCl$) and passing the liberated HCl through water.
Properties :
- It is clear, colorless and strong acidic solution.
- It have pungent odour.
- It is miscible with water and alcohol.
- On reacting with , it gives ions ($HCl + H_2O \rightarrow H_3O^+$).
Uses :
- It is used as acidifier, treat achlorhydria.
- Used as pharmaceutical aids (solvents).
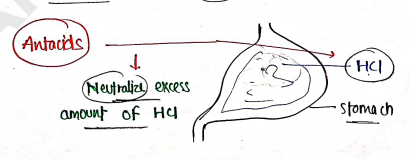
ANTACIDS
- These are those substances which are used to neutralise the excess amount of acid in stomach.
- They are mainly used in the treatment of Hyperacidity.
- Hyperacidity is the condition in which the amount of acid ($HCl$) is increases in the stomach, which can cause gastritis (inflammation in GI tract), heartburn (gastric acid enter into oesophagus through acid reflux) and peptic ulcer pain or erosion.
Ideal properties of Antacids :
- It should act rapidly for a prolonged time period.
- It should not cause constipation.
- Its pH should lie within the range of 4-6.
- It should not cause metabolic alkalosis or toxicity.
- It should not interfere with food absorption.
Mechanism : Antacids are basic in nature, which neutralise the excess amount of acid and maintain acid-base balance.
Examples :
- Sodium bicarbonate
- Aluminium and Magnesium hydroxide etc...
Classification : They are mainly divided into two parts :
- Systemic Antacids : These are those antacids which absorb in the blood circulation. These are soluble, absorbable and can cause metabolic alkalosis.
e.g. Sodium Bicarbonate.
- Non-systemic Antacids: These are those antacids which does not absorb in blood circulation instead they give locally action in stomach to decrease acidity.
e.g. Aluminium & magnesium hydroxide.
Combinations of Antacids
- It is not possible to fulfill all the criteria for an ideal antacid, so the mixtures (combination) of antacids are prepared and available in the market for desired therapeutic action of antacids.
- These combinations are mainly used to maintain the laxative effects of magnesium and the constipated effect of calcium and aluminium.
- It is also used to avoid side effects.
Examples :
- Aluminium hydroxide Gel + Magnesium Trisilicate combination It is one of the most common combinations available. It shows laxative, protective and constipated properties.
- Calcium carbonate + Antacid mixtures prolonged action with rapid onset of action.
SODIUM BICARBONATE
- It is not having less than and not more than of Sodium bicarbonate.
- Formula :
- Weight :
Method of Preparation :
When ammonium bicarbonate is react with sodium chloride, it give sodium bicarbonate (precipitate), which further separated out through filtration.
(Ammonium bicarbonate) (Sodium bicarbonate)It can also prepared through reacting Sodium Carbonate with water and carbon dioxide.
Properties :
- It is white crystalline powder with odourless, slightly alkaline taste.
- It is stable only in dry air and sparingly soluble in water and insoluble in organic solvent (e.g. alcohol, ether etc).
Assay :
- of sodium bicarbonate is weighed accurately and dissolved in of water.
- Then, the resultant mixture is titrated with using methyl orange as an indicator.
- Each ml of of .
Uses :
- Used as antacid to neutralise acid.
- Used in preparation of buffer solutions ($NaHCO_3 + H_2CO_3$).
- Used as local applicant for burns, insect bites etc.
ALUMINIUM HYDROXIDE GEL
- It should not have less than and not more than of .
- Formula :
- Weight :
Methods of Preparation: It is prepared by treating on aluminium salt (aluminium chloride) with ammonium hydroxide.
(Aluminium chloride) (Ammonium hydroxide) (Aluminium hydroxide) (Ammonium chloride)
Properties :
- It is a white colored gel, soluble in acids and alkalis.
- It is odourless and tasteless.
- Melting point is .
Uses :
- It is used as antacid to treat peptic ulcer, gastritis, gastric hyperactivity.
- To protect sun and used as a mild astringent.
MAGNESIUM HYDROXIDE MIXTURE
- It is a mixture of milk of magnesia.
- It contains not less than and not more than of .
- Molecular weight - .
Method of Preparation : When magnesium sulphate is mixed with sodium hydroxide, a magnesium hydroxide is obtained.
(Magnesium sulphate) (Sodium hydroxide) (Magnesium hydroxide)
Properties :
- It is a white coloured opaque, and more/less viscous suspension.
- It is odourless.
Uses :
- It is used as non-systemic gastric antacid and mild cathartics.
- It is less used, because on prolonged use, it can develop kidney stone.
CATHARTICS
These are those agents or drugs which are used in the treatment of constipation by facilitating defecation.
Constipation : It is a condition when a person passes less than three bowel movement (defecation) in a week or has difficult defecation.
- Defecation is the process of passing stool. It is mainly depends on peristaltic (bowel) movement i.e. movement of intestines.
- Constipation is occurs due to continuosly ignoring the urge to defecate.
- It may also occurs due to weak intestine, intestinal spasm, injury, use of certain drugs, diet etc...
- In constipation, the faecal material becomes dry and hard.
Cathartics are present into two forms:
Purgatives : These are strong cathartics, which are basically used to remove faecal material.
Laxative : These are mild acting Cathartics, which are used to treat constipation.
Classification : On the basis of their mechanism, they are classified as follow:-
- Stimulants: Those drugs which increases the bowel movement by local irritation of intestinal tract.
e.g. Senna, Aloevera, Castor oil etc..
- Bulk purgatives: As the name suggest, they increases the bulk of intestinal content which further increase the bowel movement.
e.g. Ispaghula, cellulose etc..
- Lubricants: These substance act as lubricants and increases the flow which help in emptying of bowel.
e.g. Liquid paraffin, glycerin etc.
- Saline cathartics: They increases the osmotic load of intestine which increases bowel movement. It is taken with water absorb water.
e.g. Calcium, magnesium etc.
Examples : Magnesium sulphate, sodium orthophosphate, kaolin and Bentonite.
1. Magnesium Sulphate
- It is also known as Epsom salt.
- Chemical formula:
- Molecular weight:
Method of Preparation :
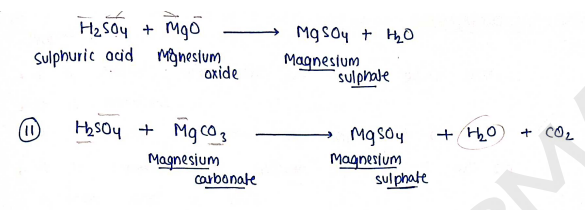
- It is prepared by neutralisation reaction b/w hot dil sulphuric acid with magnesium oxide.
Properties :
- It is white crystalline solid having a cooling saline taste.
- It is soluble in water and sparingly soluble in alcohol.
- On heating, it decomposes and evolved sulphur trioxide.
Uses :
- It is used as purgative.
- Also used as anti-convulsive agents.
2. Sodium Orthophosphate
- It is disodium hydrogen orthophosphate or sodium phosphate.
- Chemical formula: .
- Molecular Weight: .
Preparation : It is prepared by reacting sodium carbonate with a hot solution of phosphoric acid. $$Na_2CO_3 + H_3PO_4 \rightarrow Na_2HPO_4 + H_2O + CO_2$$
Properties :
- It is colorless transparent crystals of saline taste.
- It is odourless.
- It is soluble in water and insoluble in alcohol.
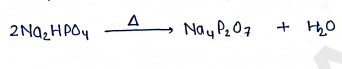
- It is converted into sodium pyrophosphate at an optimum temp of .
Uses :
- It is used as saline laxative, buffering agent or cathartics.
3. Kaolin
It is a natural, purified, hydrated aluminium silicate.
- Also known as China clay.
- Formula:
- Weight: .
- It is simply prepared by powdering, separating and purifying process of natural clay.
- It is white colored powder (clay) which is odourless & tasteless.
Uses :
- It is used as cathartics and also used in treatment of food poisoning, colitis and in cholera.
4. Bentonite
- Also known as Wilkinite.
- Formula:
- Weight: .
- It is prepared by purification of crude bentonite.
- It is an odourless, pale buff, cream coloured powder, and it is insoluble in water.
Uses :
It is used as cathartics and emulsifiers.
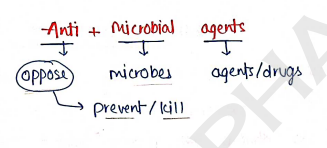
ANTI-MICROBIAL AGENTS
- These are those agents or drugs that are used to kill or inhibit the growth of microorganism.
- These are mainly work as microbicidal (to kill) or microbiostatic (prevent the growth of microbes) in nature.
Classification: These are followings:-
Antiseptic : These used mainly used to inhibit the microbial growth on living cells.
- Used in the preparation of mouthwashes, soaps, deodorants, throat & nasal sprays etc.
Disinfectants : These are those agents which kill the pathogenic microorganism to prevent infection, but these are usually applied to non-living objects.
- Mainly used to maintain hygienic conditions in home & hospitals. e.g. sulphur dioxide etc.
Germicides : Those substances which are used for both living and non-living. They also used to kill microorganism against bacteria, fungi, virus.
- Term as bactericide, fungicide, virucide etc.
Bacteriostatic : Those agents which are used to prevent the growth of bacteria. They do not kill but stop bacterial growth.
Sanitizers : These are disinfectants that are used to maintain health standards by cleaning of hands. e.g. Alcohols.
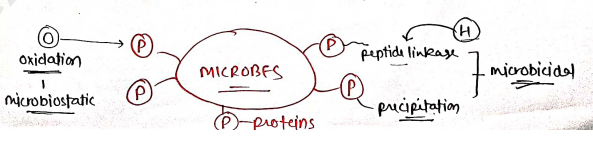
MECHANISM OF ANTIMICROBIALS
Antimicrobial agents are work by two ways:
- Microbicidal : they kill the microbes.
- Microbiostatic : they prevents the growth of microbes.
Now, Inorganic substances perform these action by three mechanism:
Oxidation : These agents oxidise the active functional groups presents in protein/enzymes which prevents the growth of microbes. (mainly applied topically). e.g. etc.
Halogenation : These agents mainly act on peptide linkage which damage the functional group present in protein results in microbial death. e.g. , etc.
Precipitation : In this, metal ions act by protein binding or protein precipitation that form complex which inactivate the protein.
EXAMPLES :
- Potassium permanganate
- Boric acid
- Hydrogen Peroxide
- Chlorinated lime
- Iodine preparations.
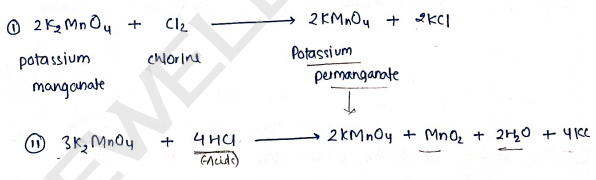
1. Potassium Permanganate [$KMnO_4$]
- It is also known as permanganate of potash or candy's crystal.
Preparation:
Properties:
- It is dark purple color.
- It is odourless with sweetish astringent like tastes.
- Soluble in water and alcohol.
Storage:
- It is stored in tightly closed containers.
Uses:
- Used as in eyewash and mouthwash.
- Used as antiseptic and as a dusting powder.
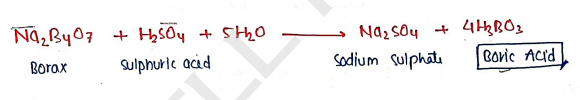
2. BORIC ACID [$H_3BO_3$]
- It is also known as hydrogen borate.
- Molecular weight: .
Preparation : It is prepared by reaction of borax with concentration sulphuric acid and followed by addition of water.
Properties:
- It is white crystalline solid.
- It is odourless with sweat taste.
- It is soluble in water and slightly soluble in alcohol.
Uses:
- It is used as in eyewash and mouthwash.
- It is also used as insecticides and bacteriostatic.
- Also used as buffer and as a dusting powder.
3. HYDROGEN PEROXIDE [$H_2O_2$]
- Mol. wt: .
- It is simplest stable peroxide also known as perhydroxide acid.
Preparation : It is prepared by the reaction of sodium peroxide with cold dil. Sulphuric acid.

Properties :
- It is clear colorless liquid.
- It is odourless having bitter acidic taste.
- It is soluble in water.
- On heating, dissociate into water & release .
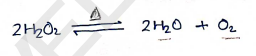
Assay : It is done by Oxidation-reduction titration.
- 10ml of the sample is diluted with 250ml of purified water.
- Take of above solution and add of Sulphuric acid ($H_2SO_4$).
- Titrated with potassium permanganate solution, until a faint pink color is obtained.
- Each ml of of .
Uses:
- It is used as disinfectant, anti-infective and deodorant.
- Also used as anticidal, germicidal, antiseptic & bleaching agents.
- Also used for tooth whitening.
4. CHLORINATED LIME [$CaOCl_2$]
- Mol. wt: .
- It is also known as Calcium oxychloride and bleaching powder.
Preparation: It is prepared by treating calcium hydroxide with Chlorine gas at about .
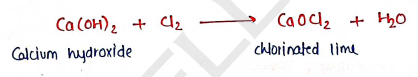
Properties:
- It is grey powder.
- Strong odour of chlorine.
- Partially soluble in water & alcohol.
Uses:
- It is used as antimicrobial, mainly bactericidal action.
- Used as bleaching agent, antiseptic, disinfectant.
- Locally used in diphtheria, scarlet fever etc...
5. IODINE ($I_2$)
- Mol. wt: .
- It is belongs to the halogens and not less than of .
Preparation: It is prepared by heating potassium iodide with Sulphuric acid and manganese dioxide.
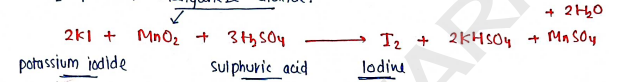
Properties:
- It is having bluish-black crystals.
- It have strong odour and volatile in nature.
- It is insoluble in water and soluble in alcohol.
Uses:
- It is used as disinfectants, antimicrobial agents.
- Used in X-ray, radiocontrast agents.
- Also used as one of the best antiseptic. e.g. Betadine.
- Iodine Preparations:
- Aqueous Iodine solution (Lugol's solution)
- Povidone Iodine
- Sodium Iodide etc. ($NaI$)
