Pharmaceutical Analysis - Unit 4
Syllabus
Redox titrations
- Concepts of oxidation and reduction
- Types of redox titrations (Principles and applications)
Cerimetry, Iodimetry, Iodometry, Bromatometry, Dichrometry, Titration with potassium iodate
Scroll to Download
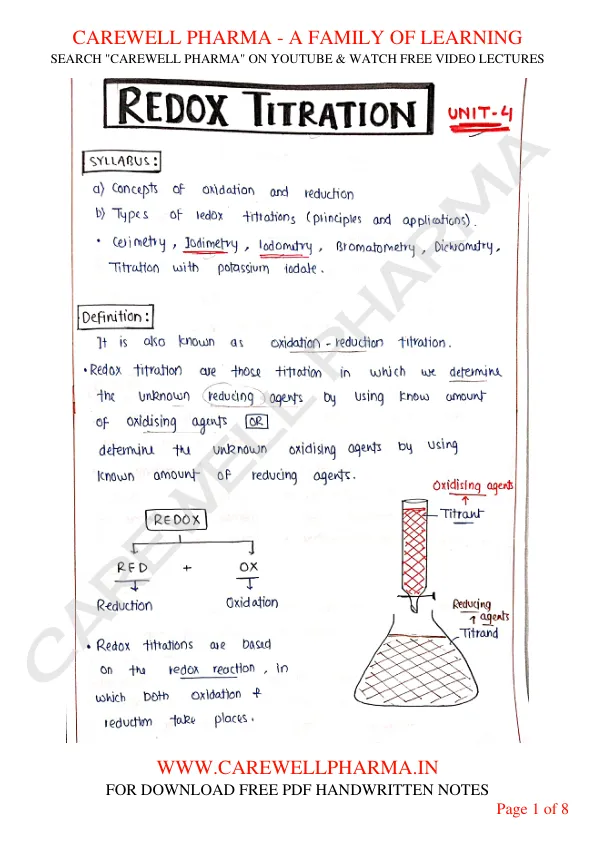
REDOX TITRATION
UNIT-4
SYLLABUS
a) Concepts of oxidation and reduction
b) Types of redox titrations (principles and applications).
Cerimetry, Iodimetry, Iodometry, Bromatometry, Dichrometry, Titration with potassium iodate.
Definition
- It is also known as oxidation - reduction titration.
- Redox titration are those titration in which we determine the unknown reducing agents by using known amount of oxidising agents. OR determine the unknown oxidising agents by using known amount of reducing agents.

- REDOX = RED (Reduction) + OX (Oxidation)
- Redox titrations are based on the redox reaction in which both oxidation & reduction take places.
CONCEPTS OF OXIDATION AND REDUCTION
- When Oxidation and reduction chemical reactions occurs together, they are termed as Redox reaction.
($Fe^{2+} \rightarrow Fe^{3+}$ Oxidation) ($Ce^{4+} \rightarrow Ce^{3+}$ Reduction)
- In this reaction, is oxidized and is reduced.
- Half reaction: (oxidation) (reduction)
OXIDATION
It is a reaction, in which loss of electrons take places and the oxidation state (valency) increases. It involves :
- Addition of Oxygen Examples:
- Removal of Hydrogen
- Loss of electrons (LEO)
REDUCTION
It is a reaction, in which gain of electrons take places and the valency decreases. It Involves:
- Removal of Oxygen
- Addition of Hydrogen
- Gain of electrons (GEO)
- Those substances which involves in oxidation-reduction (redox) reaction are known as oxidising/reducing agents.
Oxidising Agents
These are those substances which reduce itself (gain electrons) and oxidise others. Also known as Oxidants.
eg : Potassium dichromate ($K_2Cr_2O_7$), Potassium permanganate ($KMnO_4$), Potassium iodate ($KIO_3$), Hydrogen peroxide ($H_2O_2$) etc..
Reducing Agents
- These are those substances which oxidised itself (loss electrons) and reduce others. Also known as reductants.
- eg Ferrous thiosulphate, ferrous sulphate, oxalic acid etc..

REDOX INDICATORS
These are those indicators which are used in redox titration.
These are of followings
i) Internal indicator
ii) Self indicator
iii) External indicator
iv) Instrumental indicator
1. Internal indicator : Also known as those indicator which exhibit different color in oxidised and reduced form. mixed in titrand.
- Ferroin [Pale-blue (ox) Red (red)]
- Starch Iodide [Blue Colorless]
2. Self indicator : These are those in which titrant itself act as Indicator and changes the color of titrand after end point.
- eg Potassium permanganate [$KMnO_4$] (pink color), Iodine [Brown].
3. External Indicator These are those indicators which are not added in titrand instead the few drops of solution is taken periodically and mixed with Indicator solution and check the color change.
- eg Potassium ferricyanide [$K_3Fe(CN)_6$]
4. Instrumental technique: In this method we use potentiometer and conductometer instrument for the determination of end point. It is used when above three are not work.
- eg. Potentiometry.
TYPES OF REDOX TITRATION
They are of following types:
- Cerimetry
- Bromatometry
- Iodimetry
- Dichrometry
- Iodometry (Most imp.)
- Titration with potassium Iodate
IODIMETRY AND IODOMETRY
These are those titration which involves iodine. In this titration, Iodine is used as an oxidising agents.
1. IODIMETRY
- It is one type of redox titration which involves the direct titration with (Iodine).
- It is mainly based on the reaction in which Iodine is converted into iodide. (act as oxidising agents)
Principle
- In this, a standard solution of iodine ($I_2$) is used for the determination of reducing agents such as Sodium thiosulphate ($Na_2S_2O_3$), Arsenious acid ($H_3AsO_3$) etc...
- End point is determined by using starch indicator as color changes from blue to colorless.
Procedure
- Firstly we take iodine ($I_2$) in conical flask and mixed with potassium iodide ($KI$), then make up volume by adding water.
- Now, add starch as an indicator, make iodine starch solution which appear as blue color.
- Now this starch solution ($I_2$-starch Complex Blue color) is then titrated against sodium thiosulphate ($Na_2S_2O_3$) reducing agents that is filled in burette. (Iodine) (Sod. thiosulphate) (Sodium Iodide)
- Now, during titration, when all the is used from $I_2$-starch complex, the blue color start disappearing and at end point it become colorless.
Applications
- It is used for the determination of sulphur dioxide and reducing agents.
2. IODOMETRY
- It is one type of redox titration which involves indirect titration with .
- It is based on the reaction in which iodide ($I^-$) is oxidised into (Iodine) which further titrated. (Iodide) (Iodine)
Principle
In this, a standard solution of sodium thiosulphate is used for the determination of liberated iodine from potassium iodide ($KI$).
Then this iodine ($I_2$) make complex with starch solution and appear blue color.
Procedure
- Firstly take Potassium iodide ($KI$) in flask in excess amount, then add oxidising agents ($MnO_2$) which reacts with in the presence of acidic medium, which liberated Iodine. (Manganese oxide) (Iodine)
- Then add starch solution as an Indicator which forms starch-$I_2$ complex of blue color.

- Then this is titrated against standard thiosulphate solution (reducing agents) filled in burette, which converts iodine solution into iodide.
- Then after using all , color changes from blue to colorless indicates end point.
Application
- This method is mainly used for determination of both reducing as well as oxidising agents, due to its sensitivity.
CERIMETRY
- In this titration, Ceric sulphate ($Ce^{4+}$ ion) used as an oxidising agents which is basically used for the determination of ferrous ions (Fe^{2+}$), $Cu^{2+} etc.. by using ferroin as indicator.
BROMATOMETRY
- In this, Potassium bromate ($KBrO_3$) is Used as oxidising agents.
- It is estimated by adding Hydrochloric acid ($HCl$).
DICHROMETRY
- In this, potassium dichromate ($K_2Cr_2O_7$) is used as a strong oxidising titrant and act used for determination of iron salts.
TITRATION WITH POTASSIUM IODATE
- In this, potassium iodate ($KIO_3$) used as oxidising agents.
NERNST EQUATION
- It is relation between Potential of a non standard electrochemical cell and concentration of solution.
(Equilibrium Constant)
where, $E =$ Electrode potential $E^{\circ} =$ standard electrode potential $R =$ Universal gas constant $T =$ Absolute temperature $n =no. of $e^- transfer $F =$ Faraday constant.